Navigation
Install the app
How to install the app on iOS
How To Use Progressive Web App aka PWA On 420 Magazine Forum
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Making Your Own Nutrient Concentrates
- Thread starter Skybound
- Start date
- Thread starter
- #302
Skybound
Well-Known Member
Quick question.
Do we add chelated elements not in the substance list the same way as we would any other?
For eg. HB does not have copper pr Magnesium EDTA
I do. I verify that any substance in HB's database matches what my product label says. If it doesn't match, I create a Custom Solution with the correct numbers. My Cal Nite for example is a special mix and has more calcium and less ammonium than other Cal Nites.
Thanks again Sky.
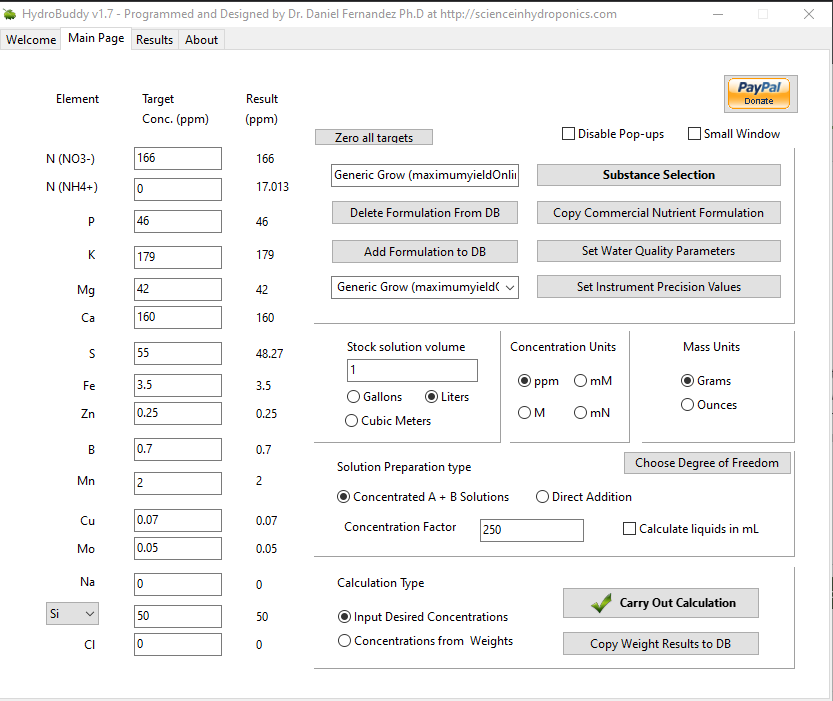
Hope its ok to ask questions as they pop up. Im playing around with a grow formula from maximumyeild.com (its got higher ppm targets than the generic grow in HB) Is this a decent recipe to start with ? I added 50ppm Si taking a clue from your posts and reading that Silicon helps with uptake of nutes. But I don't know any better.
I after a lot of trial and error, playing around with substances, bearing in mind whats available to me, got this :
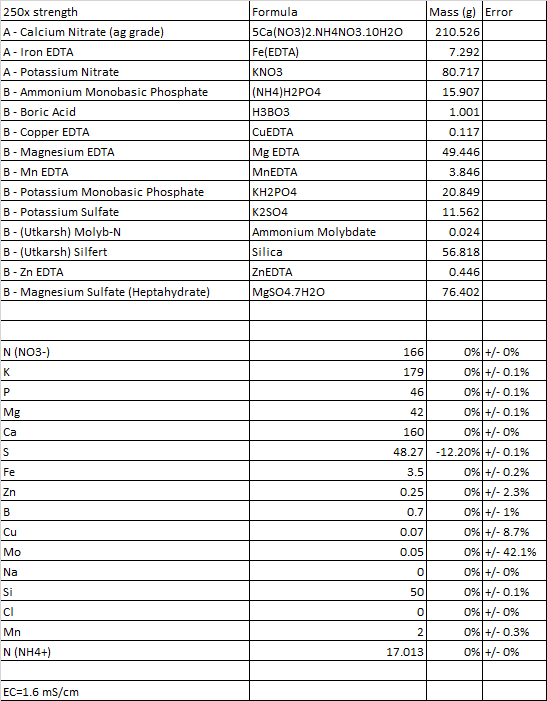
Im still getting my head around how HB calculates values. Clearly its all very interdependent. But for a moment i found myself in a situation where the app was reporting that 'Potassium sulphate' is not being used. However, if I were to add 'Mono Ammonium phosphate' to the substances, I then get a report saying that 'MAP' is not used in the formulation, but HB then goes ahead and uses Potassium sulphate.. ??? Why would the app only use Potassium Sulphate if MAP is part of, but not used, substances ? I hope im making some sense... Im fairly certain its user error, unless the app calculations take into account the sequence of substances added. I doubt...
I also have Calcium EDTA available, but adding that messes up the results.
Potassium sulphate always seems the first to go, not used in calculation, I need to understand why. For example, If I add Calcium EDTA to the substances, Potassium sulphate is no longer used, I fail to see the correlation. Is it because of incompatibilities ?
Second, I cannot get 'Sulphur' to meet targets. @ -12 its the absolute best result i have seen.
What is instrumental error ? I think its means accuracy of the EC pen, but sometimes I see 75% for Mo ?? Is it relevant ?
im going to try and read up on Sulphates vs nitrates vs chelates. But now I have taken delivery of a grow tent and a quantum light, and am really eager to mix some nutes up and get going...
Thanks again Sky, and everyone lurking!
Hope its ok to ask questions as they pop up. Im playing around with a grow formula from maximumyeild.com (its got higher ppm targets than the generic grow in HB) Is this a decent recipe to start with ? I added 50ppm Si taking a clue from your posts and reading that Silicon helps with uptake of nutes. But I don't know any better.
I after a lot of trial and error, playing around with substances, bearing in mind whats available to me, got this :
Im still getting my head around how HB calculates values. Clearly its all very interdependent. But for a moment i found myself in a situation where the app was reporting that 'Potassium sulphate' is not being used. However, if I were to add 'Mono Ammonium phosphate' to the substances, I then get a report saying that 'MAP' is not used in the formulation, but HB then goes ahead and uses Potassium sulphate.. ??? Why would the app only use Potassium Sulphate if MAP is part of, but not used, substances ? I hope im making some sense... Im fairly certain its user error, unless the app calculations take into account the sequence of substances added. I doubt...
I also have Calcium EDTA available, but adding that messes up the results.
Potassium sulphate always seems the first to go, not used in calculation, I need to understand why. For example, If I add Calcium EDTA to the substances, Potassium sulphate is no longer used, I fail to see the correlation. Is it because of incompatibilities ?
Second, I cannot get 'Sulphur' to meet targets. @ -12 its the absolute best result i have seen.
What is instrumental error ? I think its means accuracy of the EC pen, but sometimes I see 75% for Mo ?? Is it relevant ?
im going to try and read up on Sulphates vs nitrates vs chelates. But now I have taken delivery of a grow tent and a quantum light, and am really eager to mix some nutes up and get going...
Thanks again Sky, and everyone lurking!
Sky, you said :
Is this true for all Sulphates ? Then what is the source of Sulphur ? I see you using Potassium Sulphate in your MC bloom formula.
Which would mean that if I were to be using Epsom and Potassium sulphate, as I have in the above calculation, i really cant be making concentrates for long term storage ? It would need to be direct addition ?
Speaking of your MC Bloom formula, i attempted to copy it. Fine tuned the mass, Calcium Chelated came in handy here!
Ok to assume that NO3- and NH4+ cancel each other out for the correct amount of N ?
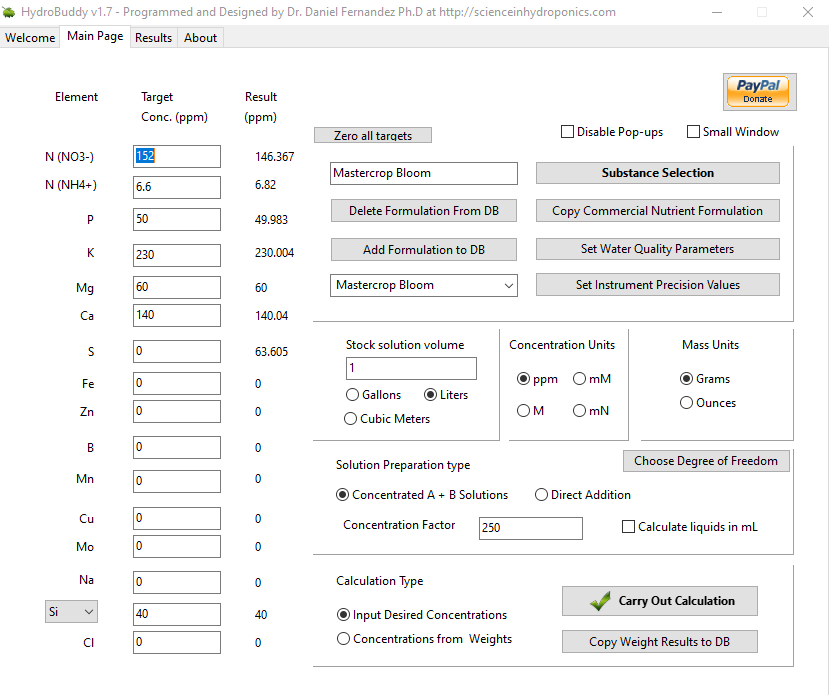
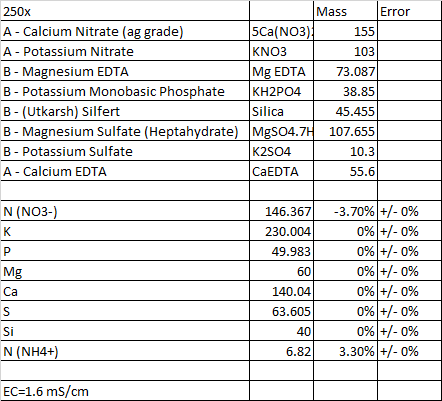
However, on the results page, why is it not mentioning the dilution levels to attain EC 1.6 ?
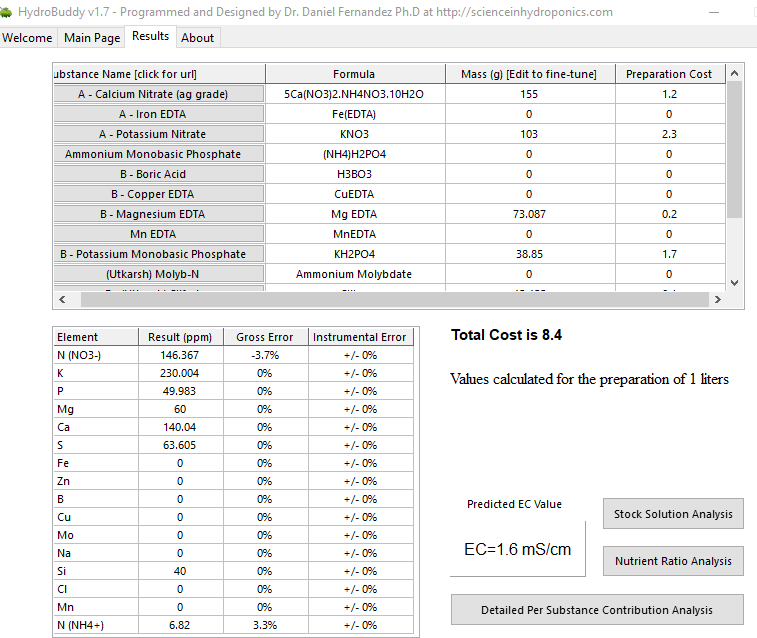
For now.
Thanks a ton!
Sulfates can be used in hydro, but they must remain in powder form until mixed, because if you try to mix them into concentrates, the heavy metals that are bonded to the sulfate molecule will quickly oxidize and turn the concentrate into rust water
Is this true for all Sulphates ? Then what is the source of Sulphur ? I see you using Potassium Sulphate in your MC bloom formula.
Which would mean that if I were to be using Epsom and Potassium sulphate, as I have in the above calculation, i really cant be making concentrates for long term storage ? It would need to be direct addition ?
Speaking of your MC Bloom formula, i attempted to copy it. Fine tuned the mass, Calcium Chelated came in handy here!
Ok to assume that NO3- and NH4+ cancel each other out for the correct amount of N ?
However, on the results page, why is it not mentioning the dilution levels to attain EC 1.6 ?
For now.
Thanks a ton!
- Thread starter
- #305
Skybound
Well-Known Member
Thanks again Sky.
Hope its ok to ask questions as they pop up. Im playing around with a grow formula from maximumyeild.com (its got higher ppm targets than the generic grow in HB) Is this a decent recipe to start with ? I added 50ppm Si taking a clue from your posts and reading that Silicon helps with uptake of nutes. But I don't know any better.
Silica aids in cell elasticity and also resistance to disease. IDK if it aids in uptake any though. In hydro, it's not such a concern as all the elements are in ionic form and immediately available for uptake. Soil elements on the other hand require the beneficial bacteria to liberate the elements and feed them to the roots in exchange for the root sugars (exudates). Farside uses 50ppm of Si, I only use 20 in veg and 40 in bloom.

I'm concerned about your ammonium content (NH4) in the above targets. That is roughly 10% of the total N and I've learned about a year ago that 10% of N as NH4 is too much. Megacrop has theirs at 5% which is what I now use. Perhaps 7% or 8% might be good too, but 10% is just a little too racey in my experience.
I after a lot of trial and error, playing around with substances, bearing in mind whats available to me, got this :


Im still getting my head around how HB calculates values. Clearly its all very interdependent. But for a moment i found myself in a situation where the app was reporting that 'Potassium sulphate' is not being used. However, if I were to add 'Mono Ammonium phosphate' to the substances, I then get a report saying that 'MAP' is not used in the formulation, but HB then goes ahead and uses Potassium sulphate.. ??? Why would the app only use Potassium Sulphate if MAP is part of, but not used, substances ? I hope im making some sense... Im fairly certain its user error, unless the app calculations take into account the sequence of substances added. I doubt...
In my experience, the app considers your targets and the solutions you have listed and tries to nail every target (except S), but if any of the elements can't be resolved as per the targets, you'll get the instrument error alerting you how much the deviation is. You'll get less deviation by expanding your salt collection. the more you give the app to work with, the easier it will be for the app to resolve the targets with zero error.
I also have Calcium EDTA available, but adding that messes up the results.
Be careful the CaEDTA doesn't have any sodium in it. I got some disolvine that's loaded with the sodium and to use the product would be toxic to my plants, so I have no use for it. AFAIK, calcium doesn't 'need' to be chelated, just as long as there isn't too much P present, the calcium won't likely bond with anything and fall out.
Potassium sulphate always seems the first to go, not used in calculation, I need to understand why. For example, If I add Calcium EDTA to the substances, Potassium sulphate is no longer used, I fail to see the correlation. Is it because of incompatibilities ?
Sulfate cannot be targeted. I wish it could, but that's not the way the app was designed for reasons unknown to me. The creator Daniel Fernandez is a botanist by profession, but he also built the app himself so he has some coding experience, but I suspect not enough to make the app very well thought out in some ways. That said, you have to mix and match different solutions to see what you can come up with. If the app says it's not using a solution, you must remove that solution from the list or for some reason, merely having it in the list skews the results. For some regimens, certain solutions just aren't used.
Second, I cannot get 'Sulphur' to meet targets. @ -12 its the absolute best result i have seen.
What is instrumental error ? I think its means accuracy of the EC pen, but sometimes I see 75% for Mo ?? Is it relevant ?
The EC pen from this point forward is only used to monitor any changes of your solution from when you first mixed it to a length of time later. The pen tries to calculate the TDS in the water by measuring the decay of an applied voltage, but there's just no way it can accurately know the actual ppm of the solution due to the many variances of electric influence each element has on the water, so it's more or less just used to measure what the "TDS" reads at the beginning, and at whatever intervals you want. Now that I'm in peat moss (ProMix), I simply do not use either of my meters except for when I make a cloning solution, I like to keep that shit at 5.5 for the rockwool starter cubes. Everything else, the PPM I know because I put it in there, and the PH of the water is not relavant due to the buffers in the dirt (garden lime and cal carbonate).
im going to try and read up on Sulphates vs nitrates vs chelates. But now I have taken delivery of a grow tent and a quantum light, and am really eager to mix some nutes up and get going...
Pretty much all of the words that have the "ate" suffix refer to a negative charged ion aka "anion" and are always listed second. The first listed in the name is the positive charged ion "cat-ion". So potassium sulfate, K is the cation(+) and the S is the anion(-). Sulfur makes your weed smoke taste more sweet, plus also works with magnesium to produce resin. Even though we can't target the S in the app, just knowing the results of more or less S will help us choose which solutions we'll have the app use. I know from experience that piney strains don't jive well with a super sweet smoke. I find the taste to be offputting and find that I would prefer the smoke to simply just taste piney without the loads of extra sweetness. Citrusy strains on the otherhand work fantastically with the extra S.
Thanks again Sky, and everyone lurking!
Hope its ok to ask questions as they pop up. Im playing around with a grow formula from maximumyeild.com (its got higher ppm targets than the generic grow in HB) Is this a decent recipe to start with ? I added 50ppm Si taking a clue from your posts and reading that Silicon helps with uptake of nutes. But I don't know any better.
Silica aids in cell elasticity and also resistance to disease. IDK if it aids in uptake any though. In hydro, it's not such a concern as all the elements are in ionic form and immediately available for uptake. Soil elements on the other hand require the beneficial bacteria to liberate the elements and feed them to the roots in exchange for the root sugars (exudates). Farside uses 50ppm of Si, I only use 20 in veg and 40 in bloom.
I'm concerned about your ammonium content (NH4) in the above targets. That is roughly 10% of the total N and I've learned about a year ago that 10% of N as NH4 is too much. Megacrop has theirs at 5% which is what I now use. Perhaps 7% or 8% might be good too, but 10% is just a little too racey in my experience.
I after a lot of trial and error, playing around with substances, bearing in mind whats available to me, got this :
Im still getting my head around how HB calculates values. Clearly its all very interdependent. But for a moment i found myself in a situation where the app was reporting that 'Potassium sulphate' is not being used. However, if I were to add 'Mono Ammonium phosphate' to the substances, I then get a report saying that 'MAP' is not used in the formulation, but HB then goes ahead and uses Potassium sulphate.. ??? Why would the app only use Potassium Sulphate if MAP is part of, but not used, substances ? I hope im making some sense... Im fairly certain its user error, unless the app calculations take into account the sequence of substances added. I doubt...
In my experience, the app considers your targets and the solutions you have listed and tries to nail every target (except S), but if any of the elements can't be resolved as per the targets, you'll get the instrument error alerting you how much the deviation is. You'll get less deviation by expanding your salt collection. the more you give the app to work with, the easier it will be for the app to resolve the targets with zero error.
I also have Calcium EDTA available, but adding that messes up the results.
Be careful the CaEDTA doesn't have any sodium in it. I got some disolvine that's loaded with the sodium and to use the product would be toxic to my plants, so I have no use for it. AFAIK, calcium doesn't 'need' to be chelated, just as long as there isn't too much P present, the calcium won't likely bond with anything and fall out.
Potassium sulphate always seems the first to go, not used in calculation, I need to understand why. For example, If I add Calcium EDTA to the substances, Potassium sulphate is no longer used, I fail to see the correlation. Is it because of incompatibilities ?
Sulfate cannot be targeted. I wish it could, but that's not the way the app was designed for reasons unknown to me. The creator Daniel Fernandez is a botanist by profession, but he also built the app himself so he has some coding experience, but I suspect not enough to make the app very well thought out in some ways. That said, you have to mix and match different solutions to see what you can come up with. If the app says it's not using a solution, you must remove that solution from the list or for some reason, merely having it in the list skews the results. For some regimens, certain solutions just aren't used.
Second, I cannot get 'Sulphur' to meet targets. @ -12 its the absolute best result i have seen.
What is instrumental error ? I think its means accuracy of the EC pen, but sometimes I see 75% for Mo ?? Is it relevant ?
The EC pen from this point forward is only used to monitor any changes of your solution from when you first mixed it to a length of time later. The pen tries to calculate the TDS in the water by measuring the decay of an applied voltage, but there's just no way it can accurately know the actual ppm of the solution due to the many variances of electric influence each element has on the water, so it's more or less just used to measure what the "TDS" reads at the beginning, and at whatever intervals you want. Now that I'm in peat moss (ProMix), I simply do not use either of my meters except for when I make a cloning solution, I like to keep that shit at 5.5 for the rockwool starter cubes. Everything else, the PPM I know because I put it in there, and the PH of the water is not relavant due to the buffers in the dirt (garden lime and cal carbonate).
im going to try and read up on Sulphates vs nitrates vs chelates. But now I have taken delivery of a grow tent and a quantum light, and am really eager to mix some nutes up and get going...
Pretty much all of the words that have the "ate" suffix refer to a negative charged ion aka "anion" and are always listed second. The first listed in the name is the positive charged ion "cat-ion". So potassium sulfate, K is the cation(+) and the S is the anion(-). Sulfur makes your weed smoke taste more sweet, plus also works with magnesium to produce resin. Even though we can't target the S in the app, just knowing the results of more or less S will help us choose which solutions we'll have the app use. I know from experience that piney strains don't jive well with a super sweet smoke. I find the taste to be offputting and find that I would prefer the smoke to simply just taste piney without the loads of extra sweetness. Citrusy strains on the otherhand work fantastically with the extra S.
Thanks again Sky, and everyone lurking!
- Thread starter
- #306
Skybound
Well-Known Member
Sky, you said :
Is this true for all Sulphates ? Then what is the source of Sulphur ? I see you using Potassium Sulphate in your MC bloom formula.
I misspoke there. I meant to say that metals that are bonded to sulfate will rust. So Iron sulfate, copper sulfate, Zinc sulfate or Manganese sulfate all will oxidize if mixed into a concentrate. As said above, you can still use the Metalic Sulfates, you just can't make concentrates with them.
The reason it's important to make concentrates though is to mitigate having to use the scale more than once, especially when you're trying to weigh out enough molydenum or other micro of super small quantity. It's just not practical to weigh out 0.013 grams of X every week. When using chelated metals, the app can consider 500 gallons of 0.01ppm molybdenum and the one time weight is multiples of grams, and when all are mixed into the water according to spec, you just need to draw Xml of the micro juice per each gallon of reservoir/feed.
Which would mean that if I were to be using Epsom and Potassium sulphate, as I have in the above calculation, i really cant be making concentrates for long term storage ? It would need to be direct addition ?
Speaking of your MC Bloom formula, i attempted to copy it. Fine tuned the mass, Calcium Chelated came in handy here!
Ok to assume that NO3- and NH4+ cancel each other out for the correct amount of N ?
N is the only element that has multiple sources of it, but each in different forms. The nitrate is useable immediately, but the NH4 has to be converted into NO3 inside the plant through an internal chemical process. NH4 can be very beneficial when used in the right ratio to NO3, but it can become toxic very quickly and too much will scorch the plant from the inside out. I suggest starting at 5% and over time if you feel you want to try more, try bumping it up to 6%, then 7 and marginally increase over time so not to screw your crop up. When considering N, think of a number, then apply 95% of it to be NO3 and 5% to be NH4.


However, on the results page, why is it not mentioning the dilution levels to attain EC 1.6 ?
My targets only use the macro solutions. You still have all of your micros listed which is likely causing the app some emotional distress.

For now.
Thanks a ton!
Is this true for all Sulphates ? Then what is the source of Sulphur ? I see you using Potassium Sulphate in your MC bloom formula.
I misspoke there. I meant to say that metals that are bonded to sulfate will rust. So Iron sulfate, copper sulfate, Zinc sulfate or Manganese sulfate all will oxidize if mixed into a concentrate. As said above, you can still use the Metalic Sulfates, you just can't make concentrates with them.
The reason it's important to make concentrates though is to mitigate having to use the scale more than once, especially when you're trying to weigh out enough molydenum or other micro of super small quantity. It's just not practical to weigh out 0.013 grams of X every week. When using chelated metals, the app can consider 500 gallons of 0.01ppm molybdenum and the one time weight is multiples of grams, and when all are mixed into the water according to spec, you just need to draw Xml of the micro juice per each gallon of reservoir/feed.
Which would mean that if I were to be using Epsom and Potassium sulphate, as I have in the above calculation, i really cant be making concentrates for long term storage ? It would need to be direct addition ?
Speaking of your MC Bloom formula, i attempted to copy it. Fine tuned the mass, Calcium Chelated came in handy here!
Ok to assume that NO3- and NH4+ cancel each other out for the correct amount of N ?
N is the only element that has multiple sources of it, but each in different forms. The nitrate is useable immediately, but the NH4 has to be converted into NO3 inside the plant through an internal chemical process. NH4 can be very beneficial when used in the right ratio to NO3, but it can become toxic very quickly and too much will scorch the plant from the inside out. I suggest starting at 5% and over time if you feel you want to try more, try bumping it up to 6%, then 7 and marginally increase over time so not to screw your crop up. When considering N, think of a number, then apply 95% of it to be NO3 and 5% to be NH4.
However, on the results page, why is it not mentioning the dilution levels to attain EC 1.6 ?
My targets only use the macro solutions. You still have all of your micros listed which is likely causing the app some emotional distress.
For now.
Thanks a ton!
wow.. Ok....
So what would be your recommendation for N ? To keep the NH4 in check ?
I cant find a combination that works in HB. the problem with manually editing a result is that is if a substance is excluded from the calculation, I cannot assign it to A or B solutions, so changing its mass from 0 has no bearing...
So what would be your recommendation for N ? To keep the NH4 in check ?
I cant find a combination that works in HB. the problem with manually editing a result is that is if a substance is excluded from the calculation, I cannot assign it to A or B solutions, so changing its mass from 0 has no bearing...
I misspoke there. I meant to say that metals that are bonded to sulfate will rust. So Iron sulfate, copper sulfate, Zinc sulfate or Manganese sulfate all will oxidize if mixed into a concentrate.
Sorry for being so slow. Wikipedia says Potassium and Magnesium are metals too... What am i missing ?
- Thread starter
- #309
Skybound
Well-Known Member
wow.. Ok....
So what would be your recommendation for N ? To keep the NH4 in check ?
I cant find a combination that works in HB. the problem with manually editing a result is that is if a substance is excluded from the calculation, I cannot assign it to A or B solutions, so changing its mass from 0 has no bearing...
I've never made an AB solution, I couldn't wrap my brain around it, especially since I tweek my feed so much. That said, I target my micros to very near Megacrop at 5 grams per gallon, then I tell the app I want to make 500 gallons of micros using those targets, then whatever it says to make the 500 gallons I stuff into a half gallon of RO. Beyond that, I only list the macros and target only macros. If I make changes, it is only to the macros and the micro juice stays the same till I use it all. It generally takes me 6 months to use a half gallon of micro juice and I'm feeding 9 in bloom and a half dozen in veg.
- Thread starter
- #310
Skybound
Well-Known Member
Sorry for being so slow. Wikipedia says Potassium and Magnesium are metals too... What am i missing ?
perhaps they're not heavy metals? IDK the difference other than the heavy metals that axidize need chelation to keep them protected until the plant uptakes the ions.
OK. So if I understand you correctly your regime is like an elaborate 3 part sold commonly, except for yours is 9 part which allows you exceptional tweaking possibilities.I've never made an AB solution, I couldn't wrap my brain around it, especially since I tweek my feed so much. That said, I target my micros to very near Megacrop at 5 grams per gallon, then I tell the app I want to make 500 gallons of micros using those targets, then whatever it says to make the 500 gallons I stuff into a half gallon of RO. Beyond that, I only list the macros and target only macros. If I make changes, it is only to the macros and the micro juice stays the same till I use it all. It generally takes me 6 months to use a half gallon of micro juice and I'm feeding 9 in bloom and a half dozen in veg.

So to simplify your method, I could reverse engineer something like the An 3 part range, or a better formula I can find...
Thanks sky! I'll keep posting. I'll see how it goes with the N.
- Thread starter
- #312
Skybound
Well-Known Member
OK. So if I understand you correctly your regime is like an elaborate 3 part sold commonly, except for yours is 9 part which allows you exceptional tweaking possibilities.
So to simplify your method, I could reverse engineer something like the An 3 part range, or a better formula I can find...
Thanks sky! I'll keep posting. I'll see how it goes with the N.
yeah, more or less. I blend the micros to a spec, but all of the rest of the components are made into their own concentrate that affords me the freedom to tweak every macro and secondary (except S ). But yeah, you can recreate EVERY brand assuming you have an adequate selection of salts.
Hey folks, quick question regarding understanding NPK ratios. I understand they mean the percentage of element in the mix. But often it seems to be rounded to a higher number.
I am copying this recipie from RIU, and the author says its a 3-1-4, HB reports it as 1:0.33:1.33, which ok if you multiply by 3, but why ?
Thanks ppl.
I am copying this recipie from RIU, and the author says its a 3-1-4, HB reports it as 1:0.33:1.33, which ok if you multiply by 3, but why ?
Thanks ppl.
- Thread starter
- #314
Skybound
Well-Known Member
You can make custom ratios so everything is relative to P, then you get your 1-3-4.
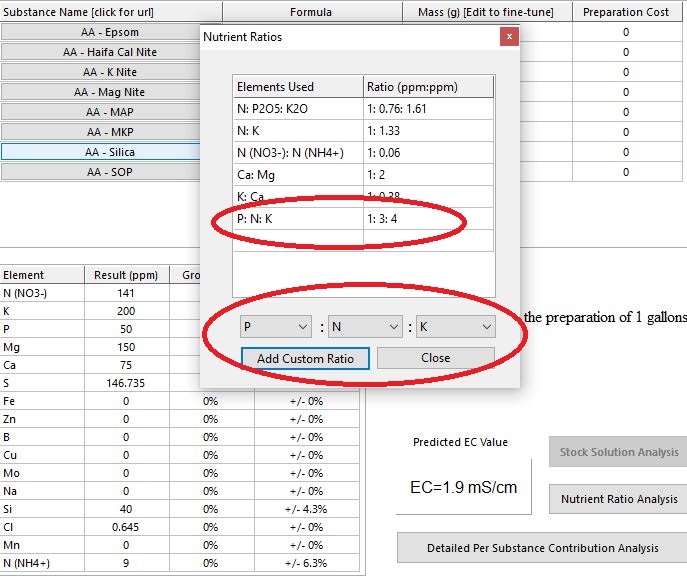
Aha! Thanks sky! Any reason why? Are NPK values a percentage of the element in the suspension (which is what I'm reading on the AN nutes) or a ratio amongst each other?
- Thread starter
- #316
Skybound
Well-Known Member
Aha! Thanks sky! Any reason why? Are NPK values a percentage of the element in the suspension (which is what I'm reading on the AN nutes) or a ratio amongst each other?
I'm uncertain why ratios are used. Perhaps it was handy for farmers 50 years ago, but all listings, even the NPK on products are listed as percentages and many people comprehend them as ratios, but you can verify that it's a percentage by matching the 3 numbers on the front label to the percentages listed in the guaranteed analysis on the back label.
Sorry for being so slow. Wikipedia says Potassium and Magnesium are metals too... What am i missing ?
There are a lot more metallic elements than most people realize. Something like 95, if I remember correctly. Also, although it's not relevant here, many elements can be considered to be metals - or not - depending on the environmental conditions (e.g., temperature and pressure). Hydrogen, for example, is generally considered to not be a metal - but you may have heard or read the term "metallic hydrogen" in the context of discussions about gas giants and such.
Thanks folks!
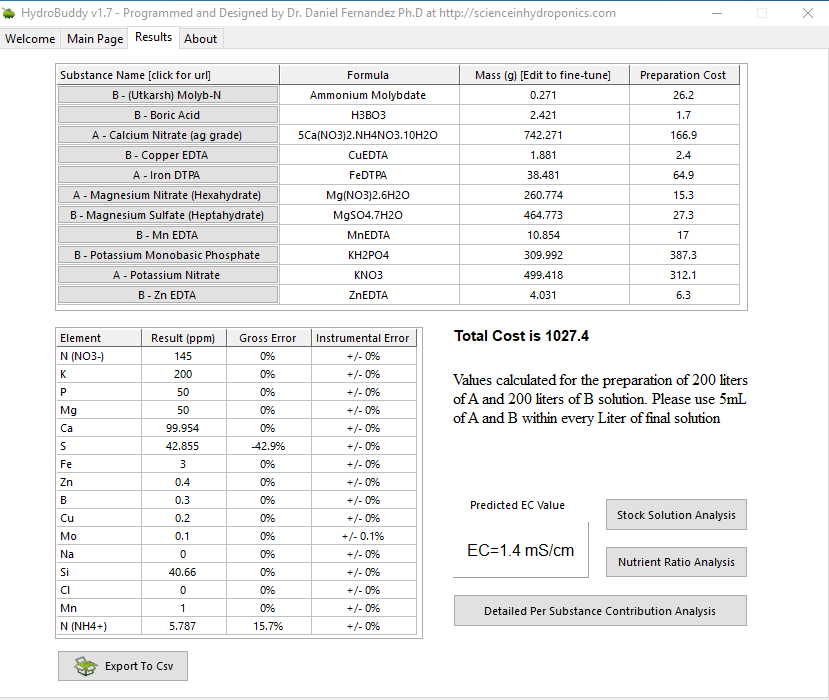
How is this looking for a complete cycle AB formula ?
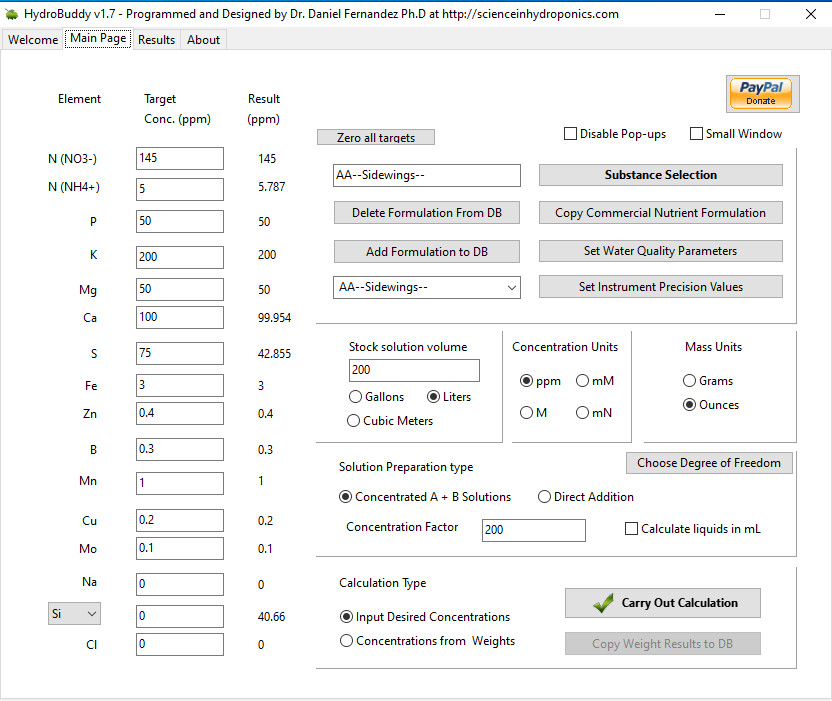
 Molyb-N is 52%Mo Ammonium Molybdate
Molyb-N is 52%Mo Ammonium Molybdate
A few questions. Is RO water from a home filter equal to Distilled water purchased for the purpose of making concentrates ? Or does distilled water have an advantage ?
REgarding CalMag, if Im using tap water, i read its probably not required. If im growing in RO water, then i would also need a CalMag additive. Why cany we just bump the CalMag values in this formula ? And how much extra CalMAg do I need if im growing in RO water ?
Thank folks!
Take care.
How is this looking for a complete cycle AB formula ?
A few questions. Is RO water from a home filter equal to Distilled water purchased for the purpose of making concentrates ? Or does distilled water have an advantage ?
REgarding CalMag, if Im using tap water, i read its probably not required. If im growing in RO water, then i would also need a CalMag additive. Why cany we just bump the CalMag values in this formula ? And how much extra CalMAg do I need if im growing in RO water ?
Thank folks!
Take care.
- Thread starter
- #319
Skybound
Well-Known Member
Thanks folks!
How is this looking for a complete cycle AB formula ?

Molyb-N is 52%Mo Ammonium Molybdate

A few questions. Is RO water from a home filter equal to Distilled water purchased for the purpose of making concentrates ? Or does distilled water have an advantage ?
REgarding CalMag, if Im using tap water, i read its probably not required. If im growing in RO water, then i would also need a CalMag additive. Why cany we just bump the CalMag values in this formula ? And how much extra CalMAg do I need if im growing in RO water ?
Thank folks!
Take care.
I'd like to see more Zinc and Boron (around 1ppm each) and as much calcium as nitrate, or marginally less. Calcium is the most finicky element in hydroponics. There are several controlable factors that can inhibit calcium uptake, the most common is being out-competed as a cation. So when there is a lot of K, Mg and NH4 to a lesser degree, less calcium is taken up and calcium is needed. Most recently I adjusted my feeds because 150-N and 130-Ca was still not enough Ca with respect to how much K and Mg I've been feeding, so I walked my feed to closer to what Megacrop is using, though IMO, the Mega uses too much K when 40-50ppm of silica is added, so I take the liberty of being able to individually lessen the K only.
The debate about RO v distilled rests solely in the TDS still in the water and I would argue personal preference. Because plants are so damn forgiving in what we feed them, I believe extreme chemical accuracy is not warranted or needed to adequately provide nutrition to plants. That said, in my grow, I have an RO unit that also deionizes and brings TDS to between 0-1ppm. I bypassed the DI stage and just run regular RO which is upwards of 20ppm/TDS. When I briefly ran the DI, I was having difficulty getting stable PH readings, so I bypassed the stage several years ago and just never reverted. That said, I don't care about the 10-20 ppm in the water that I add my salt concentrates to. It's up to you if you care about that or how much. No matter what, your concentrated salts will vastly overpower any residual TDS in whatever water you use.
About the A/B - you're on your own with that one. I know calcium isn't supposed to be mixed with P in the concentrates, but I think there are other rules that need to be adhered to to prevent precipitation. Plus also, if you find that your recipe isn't adequate, you have to toss it all out and start over. This is not the case when all of the macros are kept separate in individual concentrates.
Super thanks Skybound!
Taking your recommendation and modifying the formula gave the pleasant unexpected result of HB not using MgNitrate! This is good cause I can start immediately, MgNitrate for some reason isn't available readily, and had a 2 week wait from where I could find.
I want to keep it simple, hence the AB, pre Covid my work kept me on the road often, and I may need to have wife look into the feeding.
This looks good to you ?
No Si cause I read it raises the pH, and I dont yet have access to phosphoric or any other acid. Also read that its really not required in Hydro as all elements are readily available, think you told me... maybe..
NH4 is hopefully within safe limits also.
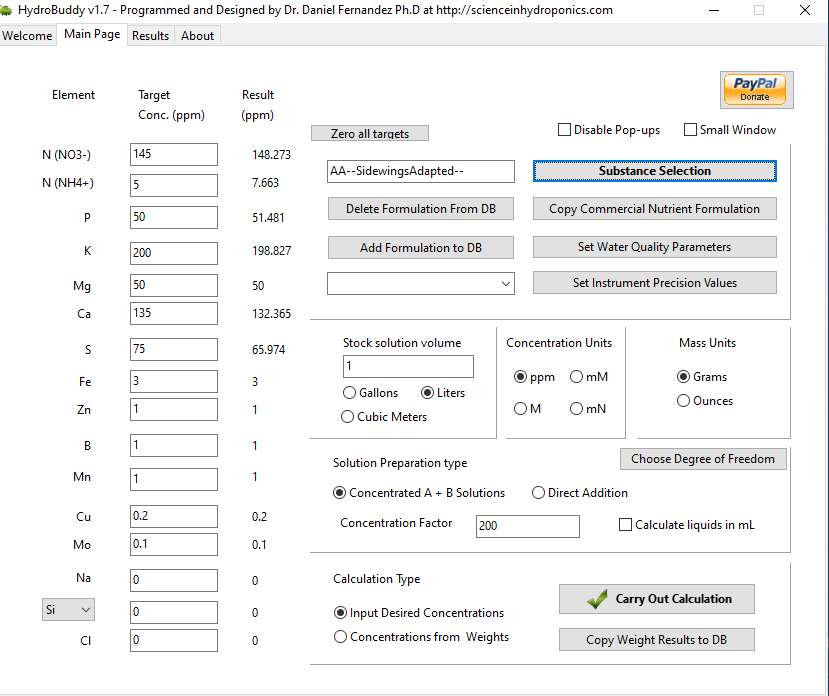
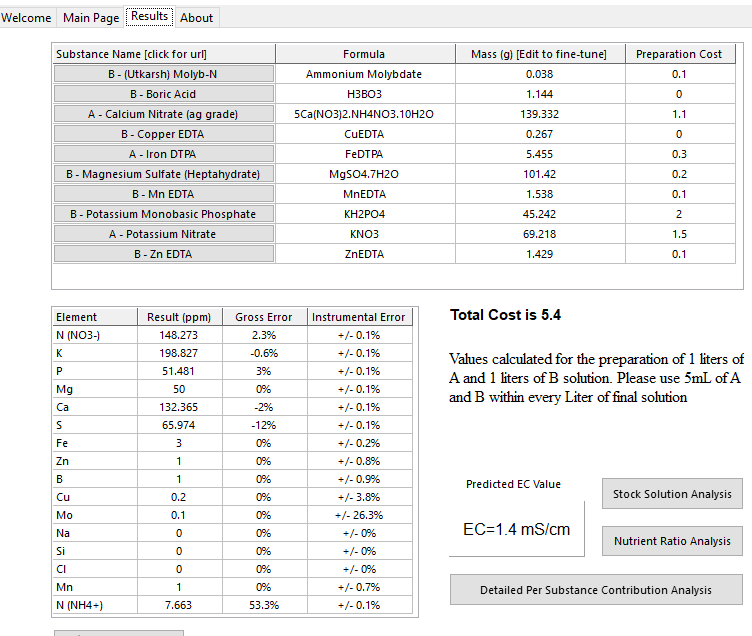
Taking your recommendation and modifying the formula gave the pleasant unexpected result of HB not using MgNitrate! This is good cause I can start immediately, MgNitrate for some reason isn't available readily, and had a 2 week wait from where I could find.
I want to keep it simple, hence the AB, pre Covid my work kept me on the road often, and I may need to have wife look into the feeding.
This looks good to you ?
No Si cause I read it raises the pH, and I dont yet have access to phosphoric or any other acid. Also read that its really not required in Hydro as all elements are readily available, think you told me... maybe..

NH4 is hopefully within safe limits also.


