I think I need a new TDS pen! 

How To Use Progressive Web App aka PWA On 420 Magazine Forum
Note: This feature may not be available in some browsers.

I think I need a new TDS pen!


Okay, I get that it’s simple tech, but doesn’t a TDS ppm 250% over elemental ppm seem a bit excessive? Mine is a $10 model LOL, and it is not possible to calibrate it.
I guess I could graph out the data and see if there’s a linear relationship between the two.

Heard an interesting logic behind this one specific to censored version of google search in China that I think escapes most ppl. So simple, yet lost on most. If they don’t work with China, China will just develop their own version of Google that competes with Google. So really, it’s the only realistic business move whether they like it, or more importantly agree with it or not. More of a passive statement, and looking forward to the calcium insights. Good work dude.I am sick of Google being caught spying and working with hostile governments
Heard an interesting logic behind this one specific to censored version of google search in China that I think escapes most ppl. So simple, yet lost on most. If they don’t work with China, China will just develop their own version of Google that competes with Google. So really, it’s the only realistic business move whether they like it, or more importantly agree with it or not. More of a passive statement, and looking forward to the calcium insights. Good work dude.
Cal (the way we want it) is a tough nut to crack, def agree on that point. I know you make all your own nutes, but did you ever check out that One Shot from NFTG? Have a friend producing some pretty insane flowers and that’s in his arsenal, so may be worth a look if you haven’t already. I grabbed some, but then switched to a prepackaged soil that’s slowly but surely destroying my garden, however One Shot is on deck for my next run. Seems like an amendment that just removes cal issues from the discussion altogether.
You know it’s probably a dumb idea, but I’m just thinking to myself why not try it. I’m using Age Old Cal (in water reservoir, I know I know) because its 20% and plant(s) love it but man does it raise pH. Counteracted by pH down = increase in EC which throws all ratios off. I’m wondering if I pour that out on a plate and let it dry/ powder out. What that can/ will do to it and it’s interaction with water pH wise. May do absolutely nothing or may surprise us. Worth a shot I suppose.
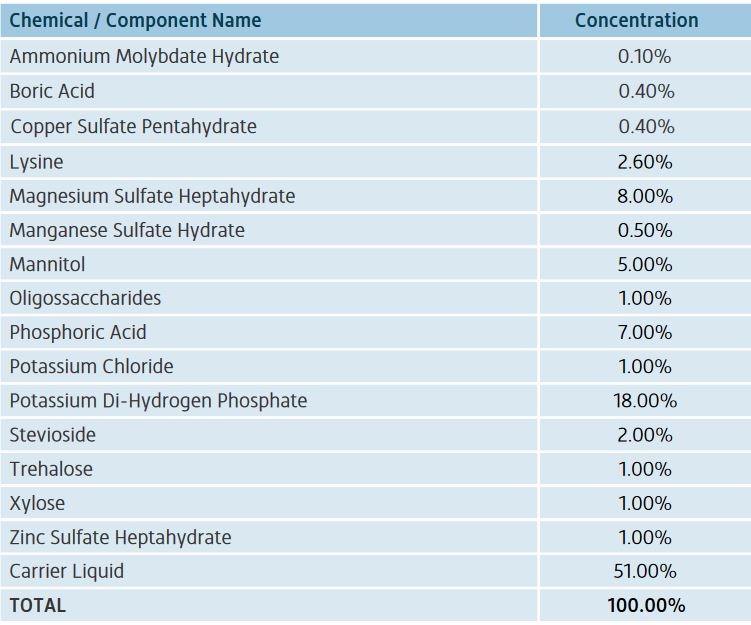
that's interesting Im waiting for you to find that golden mix im sure your going to find it to me it looks like your on to something with the foliar im hoping you find it I would like to use some of that when you find it good luck bud!After more research, I'm forming the opinion that foliar sprays containing P-K-Ca-Zn-Cu and maybe B, but also sulfur and various types of sugars, are what's needed to greatly improve brix percentages. Calcium I know is hard to move through the plant unless steady perspiration, so it is likely the primary purpose to include it in a foliar, but zinc and copper are both enzyme activators and work with Mg to create sugars. This is a product from Switzerland called VegaLab Brix Boost. I compare this product to the contents in all of Doc Bud's bottles, especially the foliar sprays as well as other notes I've taken from a hydroponics book and web searching, I can kind of see what is being applied and sort of temporarily conclude why these products are prepared this way, with these ingredients at these concentrations of each ingredient. Mannitol I just learned is a sugary alcohol that is used also as a chelator, and since I often read about chelators, that kind of info slips right into my consciousness w/o too much resistance. I don't anticipate putting together a spray worthy of testing as I want to read a good bit more on this topic and directly related tangents, but I feel like I'm onto something here.
Regarding my search for more calcium, I am next going to try adding a half gram per gallon of crushed gypsum on top of my low calcium feed regimen. Gypsum is well soluble at that concentration, and if it doesn't take well to feed the plants, I believe I'll know in a week or two. The lack wouldn't cause a deficiency, but I believe I'm already riding that line as is, so if things stay the same, good, if they improve, I think I'll notice and be able to conclude.

just carefully spray the fans and other larger leaves!Now believing that these heavy metals (in trace concentrations of course) are needed to expedite photosynthesis, assimilation of sugars and CO2, I still find myself having reservations about applying heavy metals in the regions where buds will or currently are, forming bud sites. Even though the contents of foliar sprays are generally assumed to all permeate into the leaf tissue, I still have concerns with residual traces of metallic contents in bud tissue.
I clearly have much more to learn.

Couldn't you wash off what doesn't get absorbed by the leaves?
