SmokzAlot
New Member
A compilation of material answering most questions regarding soft water and gardening. Let's first understand what we have.
Basics
The pH of tap water will vary from 5.5 to 8.5. For practical purposes the lower the pH the softer the water and the higher the pH the harder the water. Chlorine affects the pH, so let it dissipate before testing. Before adjusting the pH, add any necessary nutes noting this will more than likely affect the pH as well. PPM measures total dissolved solids within the sample. It can range from 0 to 200 on average but can go well over 500ppm which is very hard.
WIKI says "Water softening is the reduction of the concentration of calcium, magnesium, and certain other metal cations in hard water. These "hardness ions" can cause a variety of undesired effects including interfering with the action of soaps, the build up of limescale, which can foul plumbing, and galvanic corrosion.[1] Conventional water-softening appliances intended for household use depend on an ion-exchange resin in which hardness ions are exchanged for sodium ions. Water softening may be desirable where the source of water is hard.[2] However, hard water also conveys some benefits to health by providing dietary calcium and magnesium and reducing the solubility of potentially toxic metal ions such as lead and copper[3]."
How Water Is Softened
The most common method used for softening water is ion exchange, where the dissolved calcium and magnesium ions are exchanged for sodium or potassium ions. Sodium and potassium do not cause the problems that are associated with calcium and magnesium. The process is the basis for most water softening equipment on the market today.
Water enters a water softener where it comes in contact with a bed of tiny beads that hold sodium chloride or potassium chloride ions. Since the beads are chemically more attracted to calcium and magnesium ions, ion exchange occurs. The calcium and magnesium ions "stick" to the surface of the beads, dislodging the sodium or potassium. After the beads are completely exhausted, a system then regenerates and replaces the exhausted sodium or potassium in the field. After the extra solution is rinsed from the resin bed, the entire ion exchange cycle begins again.
So where do we stand at this point? We now have a salt heavy water along with calcium and magnesium deficiencies. We must also be aware that our PPM will remain near the same. The process as stated replaces one for the other so dissolved solids don't change so much. In reality all will fluctuate some. More so in a well water system. As they are directly impacted by environmental changes such as heavy rainfalls and floods, as well as drought. In my opinion to have any success in gardening you need to work with a consistent water source more so that proper. Adjustments can be made but keeping up with variables can be a pain. Here is where I think RO water comes in. It does strip many things form the water, but you now have a known and consistent water source. Here is where each grower must decide what is feasible, efficient, and affordable for their environment. A reasonable goal would be to minimize the issues you deal with if you cant eliminate them. My logic would then dictate that one is better off in most places to use the hard water instead. Use the beneficial calcium and magnesium and avoid the potential of salt lock up. Again this must be weighed in against medium choices, watering styles, and availability. Example: It is my understanding that coco will hold salts very easily as this is it's genetic design and purpose. It is also my understanding that you can deal with this with frequent watering, it will help keep levels down. Example: Less nutes (salts) and more frequency. End result still getting the nutes just not at once. Always watering to waste seems to be very beneficial for that medium and salt retention. So is it possible that soft water may have less of an impact with coco and frequent watering to waste? Perhaps someone can verify that as I have not tried it yet.
What is pH?
A measure of the acidity or alkalinity of a solution, numerically equal to 7 for neutral solutions, increasing with increasing alkalinity and decreasing with increasing acidity.
What is PPM?
PPM is very easily defined as Parts Per Million and can be used as the measurement of a number of different things. Example: In hydroponics, the measurement is used to measure the amount of Total Dissolved Solids in your nutrient solution.
What is EC?
EC is a unit of measure to gauge the Electrical Conductivity of a solution. An EC meter applies an electrical voltage to the solution and reads the conductivity that is produced from the motion of mineral Ions. A lot of meters today do the conversion for you to ppm. Commonly referred to as a TDS/EC meter.
Is your water hard?
How is hardness measured?
There are two units commonly used in measuring hardness: German Degrees of Hardness (dH) and Parts Per Million (ppm). One dH = 17 ppm. For a rule of thumb:
0 to 4 dH - 0 to 70 ppm - very soft
4 to 8 dH - 70 to 125 ppm - soft
8 to 12 dH - 125 to 200 ppm - medium hard
12 to 18 dH - 200 to 300 ppm - hard
18 to 30 dH - 300 to 500 ppm - very hard
over 30 dH over 500 ppm - extremely hard
Without getting into a PH and PPM/EC discussion one needs to understand the water they have available and either make the best of it or improve it. One can use RO, RW, DS, or a mix of. Know what you have for a base, it's as important as knowing the PH and total alkalinity. With as stable/consistent water supply one can devise a plan of dealing with it. Will you need to supplement cal/mag? Will you need to flush more? Just how hard is your water? In my mind I would rather deal with changing cal/mag levels than salt levels. Rainy season drives the ppm count up here which tends to equate to more salt as well. Causing more issues, and making it more difficult to identify deficits.
Hard Water Effects
Hard water reduces the control the gardener has over the medium acidity, which is a bad thing, but the water itself may not always be bad for the garden. Whats the Ph? What is the total alkalinity?
Hard Water and pH
Hard water contains calcium carbonate and magnesium carbonate, the same chemicals found in limestone. Limestone is often used in small amounts to raise the pH of your garden, and hard water can do the same thing. The amount of difference the hard water makes depends on how much you use and how hard the water is. Hard water typically has its biggest impact with more frequent watering gardening styles.
What are others saying?
"Is softened water harmful to plants, lawns, and gardens?
Softened water is not recommended for watering plants, lawns, and gardens due to its sodium content. Care must also be taken that water used in recharging a water softener be disposed of through a storm drain or sewer because of its damaging effects. If you are on a septic tank, the logical method of disposal is to discharge the brine into the septic tank and soil absorption field where some leaching of sodium salts will occur. Other alternatives include a separate holding tank, which could be emptied by a vacuum truck, a separate disposal field, or discharge point that does not affect neighbors' property." -University of Kentucky College of Agriculture
"Dissolved salts caused by saline water and fertilizer build up to toxic levels quickly in container gardens. Excessive salts inhibit seed germination, burn the root hairs, and tips or edges of leaves and stunt the plant. Flush excess salt built up from growing mediums by applying 2 gallons of water per gallon of medium. Repeat leaching using a mild pH-corrected fertilizer solution. Leach growing medium every 2 — 4 weeks if using soft water or saline water. Hard water and well water in dry climates is often alkaline and usually contains notable amounts of calcium and magnesium. Cannabis uses large quantities of both nutrients, but too much calcium or magnesium can build-up in soil. In general, water that tastes good to people, also "tastes" good to cannabis."
"The concentration of calcium and magnesium indicate how "hard" the water is. Water containing 100 to 150 milligrams of calcium (CaCO3) per liter is acceptable to grow marijuana. "Soft" water contains less than 50 milligrams of calcium per liter and should be supplemented with calcium and magnesium.
At low levels, sodium appears to bolster yields, possibly acting as a partial substitute for potassium deficiencies. But when excessive, sodium is toxic and induces deficiencies of other nutrients, primarily potassium, calcium and magnesium." -Jorge Cervantes
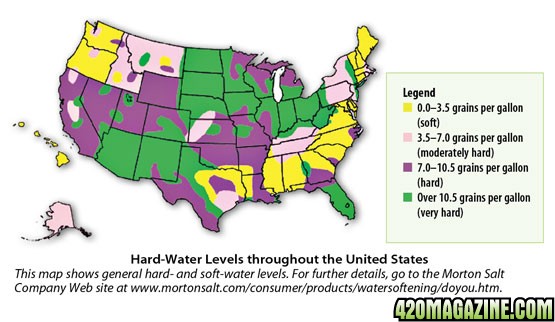
George Elliot, an associate professor of horticulture specializing in plant nutrition and water quality at the University of Connecticut says: "Water hardness and softness can have a significant effect on plants, especially those growing in containers. Hardness is related to the content of calcium carbonate and magnesium carbonate dissolved in the water, often expressed in units of grains per gallon. Hard water has from 7.0 to 10.5 grains, and very hard water has in excess of 10.5 grains. You may recognize that calcium carbonate and magnesium carbonate are the components of limestone. When hard water is used for irrigation, it's the same as adding a small amount of lime every time you water. Over time, this continual addition of lime will increase the pH of the growing medium.
An increase in pH is more pronounced in containers because they are watered frequently. Soils, however, are not as affected because rainfall helps to counteract it. Nevertheless, hard water can still cause some problems when irrigating outdoors. In overhead sprinklers, it can cause white, scalelike deposits to develop on leaves as minerals are left behind when the water evaporates. Similar deposits can clog drip-irrigation systems.
Many plants can tolerate a wide range of pH, although some plants–most notably petunias and acid-loving plants like azaleas–are sensitive to high pH levels. An increase in pH causes young leaves to turn pale and yellowish, while leaf veins remain green. This interveinal chlorosis is sometimes referred to as a lime-induced chlorosis and is actually the result of an iron deficiency due to high pH levels. It won't kill the plant, but it will make it look unhealthy and reduce its vigor.
Hard water can be found in large areas of the United States (map, above). The only way to tell if your water is hard is to test it. Easy-to-use kits for do-it-yourself testing are available at most hardware stores.
If you have hard water, you can use a water softener to treat it for household use, but you should not use softened water for irrigation. Softened water has a high salt (sodium chloride) content, which causes its own set of problems. "
you may want to check out:
What is the difference between ppm and EC?
Basics
The pH of tap water will vary from 5.5 to 8.5. For practical purposes the lower the pH the softer the water and the higher the pH the harder the water. Chlorine affects the pH, so let it dissipate before testing. Before adjusting the pH, add any necessary nutes noting this will more than likely affect the pH as well. PPM measures total dissolved solids within the sample. It can range from 0 to 200 on average but can go well over 500ppm which is very hard.
WIKI says "Water softening is the reduction of the concentration of calcium, magnesium, and certain other metal cations in hard water. These "hardness ions" can cause a variety of undesired effects including interfering with the action of soaps, the build up of limescale, which can foul plumbing, and galvanic corrosion.[1] Conventional water-softening appliances intended for household use depend on an ion-exchange resin in which hardness ions are exchanged for sodium ions. Water softening may be desirable where the source of water is hard.[2] However, hard water also conveys some benefits to health by providing dietary calcium and magnesium and reducing the solubility of potentially toxic metal ions such as lead and copper[3]."
How Water Is Softened
The most common method used for softening water is ion exchange, where the dissolved calcium and magnesium ions are exchanged for sodium or potassium ions. Sodium and potassium do not cause the problems that are associated with calcium and magnesium. The process is the basis for most water softening equipment on the market today.
Water enters a water softener where it comes in contact with a bed of tiny beads that hold sodium chloride or potassium chloride ions. Since the beads are chemically more attracted to calcium and magnesium ions, ion exchange occurs. The calcium and magnesium ions "stick" to the surface of the beads, dislodging the sodium or potassium. After the beads are completely exhausted, a system then regenerates and replaces the exhausted sodium or potassium in the field. After the extra solution is rinsed from the resin bed, the entire ion exchange cycle begins again.
So where do we stand at this point? We now have a salt heavy water along with calcium and magnesium deficiencies. We must also be aware that our PPM will remain near the same. The process as stated replaces one for the other so dissolved solids don't change so much. In reality all will fluctuate some. More so in a well water system. As they are directly impacted by environmental changes such as heavy rainfalls and floods, as well as drought. In my opinion to have any success in gardening you need to work with a consistent water source more so that proper. Adjustments can be made but keeping up with variables can be a pain. Here is where I think RO water comes in. It does strip many things form the water, but you now have a known and consistent water source. Here is where each grower must decide what is feasible, efficient, and affordable for their environment. A reasonable goal would be to minimize the issues you deal with if you cant eliminate them. My logic would then dictate that one is better off in most places to use the hard water instead. Use the beneficial calcium and magnesium and avoid the potential of salt lock up. Again this must be weighed in against medium choices, watering styles, and availability. Example: It is my understanding that coco will hold salts very easily as this is it's genetic design and purpose. It is also my understanding that you can deal with this with frequent watering, it will help keep levels down. Example: Less nutes (salts) and more frequency. End result still getting the nutes just not at once. Always watering to waste seems to be very beneficial for that medium and salt retention. So is it possible that soft water may have less of an impact with coco and frequent watering to waste? Perhaps someone can verify that as I have not tried it yet.
What is pH?
A measure of the acidity or alkalinity of a solution, numerically equal to 7 for neutral solutions, increasing with increasing alkalinity and decreasing with increasing acidity.
What is PPM?
PPM is very easily defined as Parts Per Million and can be used as the measurement of a number of different things. Example: In hydroponics, the measurement is used to measure the amount of Total Dissolved Solids in your nutrient solution.
What is EC?
EC is a unit of measure to gauge the Electrical Conductivity of a solution. An EC meter applies an electrical voltage to the solution and reads the conductivity that is produced from the motion of mineral Ions. A lot of meters today do the conversion for you to ppm. Commonly referred to as a TDS/EC meter.
Is your water hard?

How is hardness measured?
There are two units commonly used in measuring hardness: German Degrees of Hardness (dH) and Parts Per Million (ppm). One dH = 17 ppm. For a rule of thumb:
0 to 4 dH - 0 to 70 ppm - very soft
4 to 8 dH - 70 to 125 ppm - soft
8 to 12 dH - 125 to 200 ppm - medium hard
12 to 18 dH - 200 to 300 ppm - hard
18 to 30 dH - 300 to 500 ppm - very hard
over 30 dH over 500 ppm - extremely hard
Without getting into a PH and PPM/EC discussion one needs to understand the water they have available and either make the best of it or improve it. One can use RO, RW, DS, or a mix of. Know what you have for a base, it's as important as knowing the PH and total alkalinity. With as stable/consistent water supply one can devise a plan of dealing with it. Will you need to supplement cal/mag? Will you need to flush more? Just how hard is your water? In my mind I would rather deal with changing cal/mag levels than salt levels. Rainy season drives the ppm count up here which tends to equate to more salt as well. Causing more issues, and making it more difficult to identify deficits.
Hard Water Effects
Hard water reduces the control the gardener has over the medium acidity, which is a bad thing, but the water itself may not always be bad for the garden. Whats the Ph? What is the total alkalinity?
Hard Water and pH
Hard water contains calcium carbonate and magnesium carbonate, the same chemicals found in limestone. Limestone is often used in small amounts to raise the pH of your garden, and hard water can do the same thing. The amount of difference the hard water makes depends on how much you use and how hard the water is. Hard water typically has its biggest impact with more frequent watering gardening styles.
What are others saying?
"Is softened water harmful to plants, lawns, and gardens?
Softened water is not recommended for watering plants, lawns, and gardens due to its sodium content. Care must also be taken that water used in recharging a water softener be disposed of through a storm drain or sewer because of its damaging effects. If you are on a septic tank, the logical method of disposal is to discharge the brine into the septic tank and soil absorption field where some leaching of sodium salts will occur. Other alternatives include a separate holding tank, which could be emptied by a vacuum truck, a separate disposal field, or discharge point that does not affect neighbors' property." -University of Kentucky College of Agriculture
"Dissolved salts caused by saline water and fertilizer build up to toxic levels quickly in container gardens. Excessive salts inhibit seed germination, burn the root hairs, and tips or edges of leaves and stunt the plant. Flush excess salt built up from growing mediums by applying 2 gallons of water per gallon of medium. Repeat leaching using a mild pH-corrected fertilizer solution. Leach growing medium every 2 — 4 weeks if using soft water or saline water. Hard water and well water in dry climates is often alkaline and usually contains notable amounts of calcium and magnesium. Cannabis uses large quantities of both nutrients, but too much calcium or magnesium can build-up in soil. In general, water that tastes good to people, also "tastes" good to cannabis."
"The concentration of calcium and magnesium indicate how "hard" the water is. Water containing 100 to 150 milligrams of calcium (CaCO3) per liter is acceptable to grow marijuana. "Soft" water contains less than 50 milligrams of calcium per liter and should be supplemented with calcium and magnesium.
At low levels, sodium appears to bolster yields, possibly acting as a partial substitute for potassium deficiencies. But when excessive, sodium is toxic and induces deficiencies of other nutrients, primarily potassium, calcium and magnesium." -Jorge Cervantes
George Elliot, an associate professor of horticulture specializing in plant nutrition and water quality at the University of Connecticut says: "Water hardness and softness can have a significant effect on plants, especially those growing in containers. Hardness is related to the content of calcium carbonate and magnesium carbonate dissolved in the water, often expressed in units of grains per gallon. Hard water has from 7.0 to 10.5 grains, and very hard water has in excess of 10.5 grains. You may recognize that calcium carbonate and magnesium carbonate are the components of limestone. When hard water is used for irrigation, it's the same as adding a small amount of lime every time you water. Over time, this continual addition of lime will increase the pH of the growing medium.
An increase in pH is more pronounced in containers because they are watered frequently. Soils, however, are not as affected because rainfall helps to counteract it. Nevertheless, hard water can still cause some problems when irrigating outdoors. In overhead sprinklers, it can cause white, scalelike deposits to develop on leaves as minerals are left behind when the water evaporates. Similar deposits can clog drip-irrigation systems.
Many plants can tolerate a wide range of pH, although some plants–most notably petunias and acid-loving plants like azaleas–are sensitive to high pH levels. An increase in pH causes young leaves to turn pale and yellowish, while leaf veins remain green. This interveinal chlorosis is sometimes referred to as a lime-induced chlorosis and is actually the result of an iron deficiency due to high pH levels. It won't kill the plant, but it will make it look unhealthy and reduce its vigor.
Hard water can be found in large areas of the United States (map, above). The only way to tell if your water is hard is to test it. Easy-to-use kits for do-it-yourself testing are available at most hardware stores.
If you have hard water, you can use a water softener to treat it for household use, but you should not use softened water for irrigation. Softened water has a high salt (sodium chloride) content, which causes its own set of problems. "
you may want to check out:
What is the difference between ppm and EC?


