I would like someone who makes their own fertilizer to check my math.
I've learned Hydro Buddy and I have created my "ideal" nutrient recipe (in theory).
However, I want to make sure I understand the formula for adding Disodium EDTA to chelate my Fe, Mn, Zn & Cu.
From what I have extrapolated from Dr. Daniel Fernandez's various comments at the bottom of his DIY Nutrient videos on YouTube, it goes something like this.
1) After Final Recipe has been chosen and concentrated (i.e. 1 Liter @ 200:1),
you take the millimolar concentration amount and change 'ppm' to mM' on "Main Page."
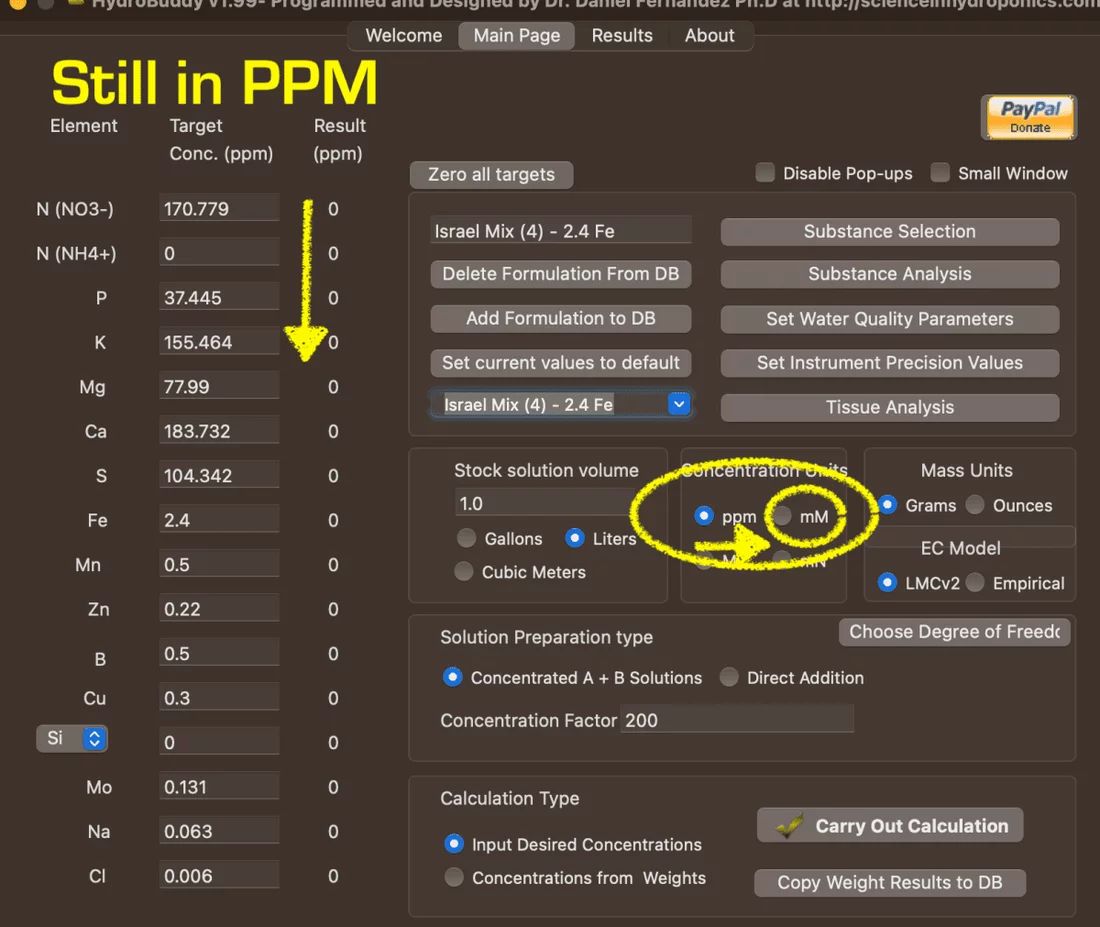
After Switching to 'mM," it looks like this:
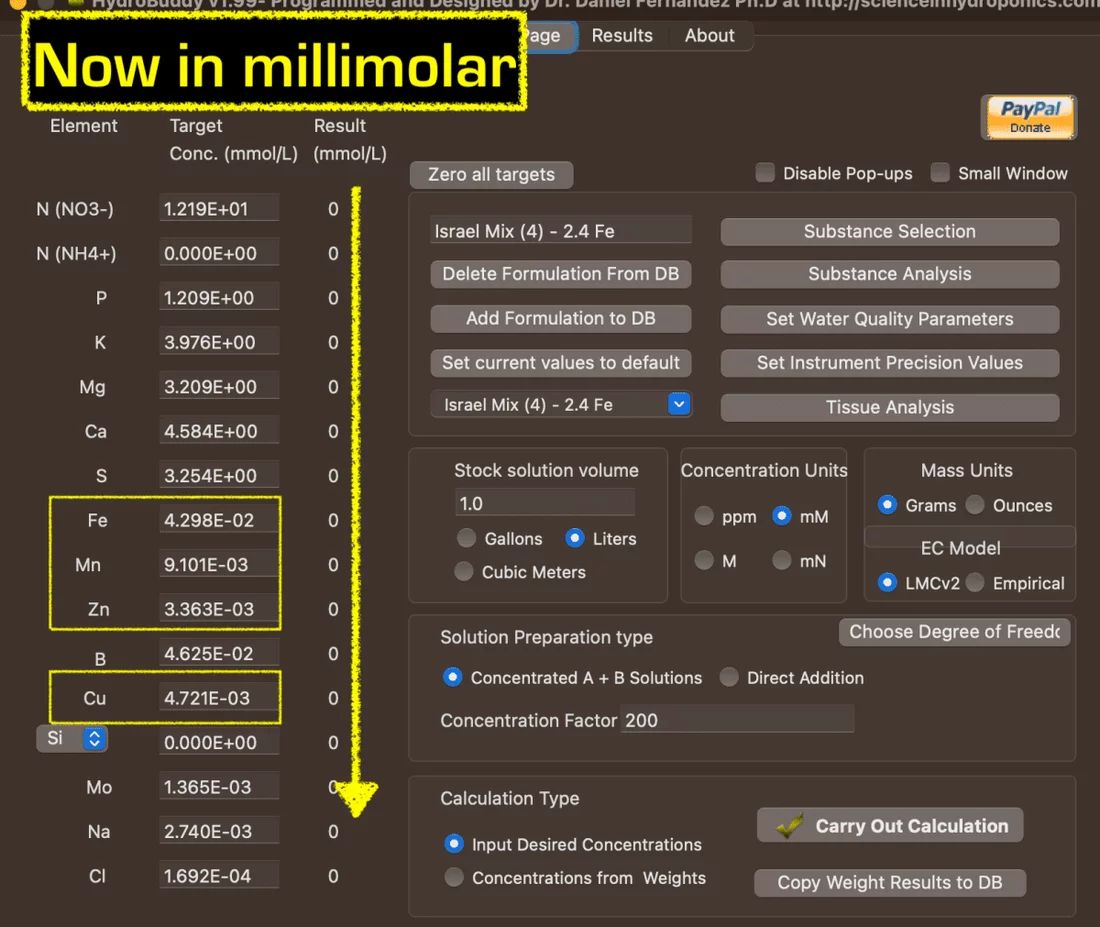
2) Once you have the millimolar concentration, you add up the 4 numbers:
Fe: 4.298E-02
Mn: 9.101E-03
Zn: 3.363E-03
Cu: 4.721E-03
-------------------
= 6.02E-02 (0.0602)
Actually it is: 0.060165
3) Once you establish this number:
Multiply by the concentration Amount (200:1). It would be multiply by 100 if 100:1.
0.060165 x 200
= 12.033
4) Next he likes to add 20 or 25% for safety.
12.033 x 1.25
=15.041
5) Now you take your "safe" total and multiply by the molar mass of Disodium EDTA (336.21) for your grams:
15.0412 x 0.33621
That leaves you with the grams required.
However, the number isn't necessairly complete.
In this case it is, but if amount was 0.5 liter it would be differnet.
For Final Number, Multiply grams by volume of concentration (1 Liter):
5.06 x 1.00
= 5.06 grams
If it it were for 0.5 liters, then it would be :
5.06 x 0.5
=2.53 grams
Below I'll show an example (not final) of my "template" worksheet:
Items in RED are to remind me to double check things that can change:
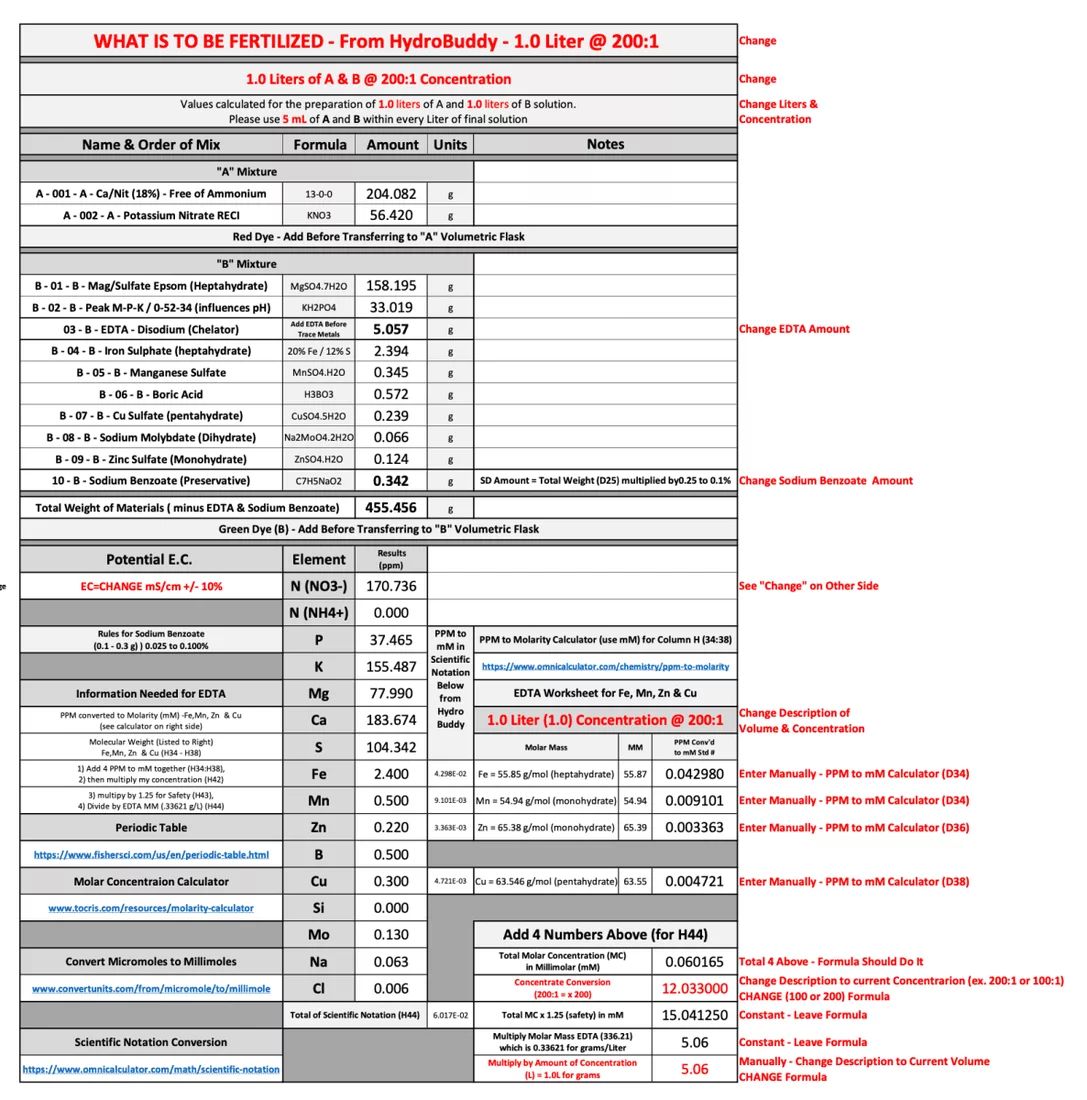
I've learned Hydro Buddy and I have created my "ideal" nutrient recipe (in theory).
However, I want to make sure I understand the formula for adding Disodium EDTA to chelate my Fe, Mn, Zn & Cu.
From what I have extrapolated from Dr. Daniel Fernandez's various comments at the bottom of his DIY Nutrient videos on YouTube, it goes something like this.
1) After Final Recipe has been chosen and concentrated (i.e. 1 Liter @ 200:1),
you take the millimolar concentration amount and change 'ppm' to mM' on "Main Page."
After Switching to 'mM," it looks like this:
2) Once you have the millimolar concentration, you add up the 4 numbers:
Fe: 4.298E-02
Mn: 9.101E-03
Zn: 3.363E-03
Cu: 4.721E-03
-------------------
= 6.02E-02 (0.0602)
Actually it is: 0.060165
3) Once you establish this number:
Multiply by the concentration Amount (200:1). It would be multiply by 100 if 100:1.
0.060165 x 200
= 12.033
4) Next he likes to add 20 or 25% for safety.
12.033 x 1.25
=15.041
5) Now you take your "safe" total and multiply by the molar mass of Disodium EDTA (336.21) for your grams:
15.0412 x 0.33621
That leaves you with the grams required.
However, the number isn't necessairly complete.
In this case it is, but if amount was 0.5 liter it would be differnet.
For Final Number, Multiply grams by volume of concentration (1 Liter):
5.06 x 1.00
= 5.06 grams
If it it were for 0.5 liters, then it would be :
5.06 x 0.5
=2.53 grams
Below I'll show an example (not final) of my "template" worksheet:
Items in RED are to remind me to double check things that can change:






