Jim Finnel
Fallen Cannabis Warrior & Ex News Moderator
Cannabinoid Receptor Ligands Part 1
Roger G Pertwee School of Medical Sciences, Institute of Medical Sciences, University of Aberdeen, Foresterhill, Aberdeen
Roger Pertwee is currently Professor of Neuropharmacology at the University of Aberdeen and Director of Pharmacology for GW Pharmaceuticals. His research focuses on thepharmacology of cannabis and its constituents and of cannabinoid receptors and cannabis-derived, synthetic and endogenous ligands for these receptors.
The Endocannabinoid System including immune cells. CB 2 receptors are located Two types of cannabinoid receptor have so far been predominantly in immune cells both within and identified. 1,2 These are the CB 1 receptor, cloned outside the central nervous system, the functions in 1990, 3 and the CB 2 receptor, cloned in 1993, 4 of these receptors including modulation of cytokine both of which are members of the superfamily of release and of immune cell migration. In the brain, G-protein-coupled receptors. The cloning of these CB 2 receptors are expressed by microglia, 7 by blood receptors prompted the development of mice from vessels, 7 and by some neurons. 8,9 However, the role which cannabinoid CB 1 and/or CB 2 receptors have of neuronal CB 2 receptors is currently unknown. been genetically deleted and these transgenic The central distribution pattern of CB 1 receptors is animals, particularly CB 1 knockout mice, are now heterogeneous and accounts for several prominent widely used to explore the physiological and pharmacological properties of CB 1 receptor agonists, pathological functions of cannabinoid receptors. 1,5,6 for example their ability to impair cognition and CB 1 receptors are found mainly at the terminals of memory and to alter the control of motor function. Thus central and peripheral neurons where they usually the cerebral cortex, hippocampus, lateral caudate- mediate inhibition of neurotransmitter release. putamen, substantia nigra pars reticulata, globus They are also present in some non-neuronal cells, pallidus, entopeduncular nucleus and the molecular
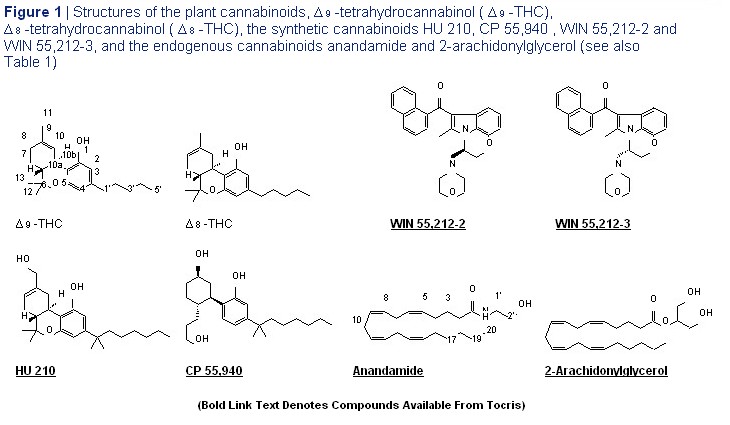
layer of the cerebellum are all populated with such endogenous cannabinoids (endocannabinoids) particularly high concentrations of CB 1 receptors. 1,10 to be identified were N -arachidonoyl ethanolamine In line with the analgesic properties of cannabinoid (anandamide) in 1992 and 2-arachidonylglycerol in receptor agonists, CB 1 receptors are also found on 1995 (Figure 1), 20-22 both of which are synthesised pain pathways in the brain and spinal cord and at the on demand in response to elevations of intracellular peripheral terminals of primary sensory neurons. 11,12 calcium. 23 Anandamideisformedfrom N -arachidonoyl Although the concentration of CB 1 receptors is phosphatidylethanolamine in a process that is considerably less in peripheral tissues than in the catalysed by N -acyl phosphatidylethanolamine- central nervous system, this does not mean that selective phospholipase D (NAPE-PLD). The peripheral CB 1 receptors are unimportant. Thus in synthesis of
2-arachidonylglycerol, however, some peripheral tissues, discrete regions such as is thought to depend on the conversion of nerve terminals that form only a small part of the
2-arachidonate-containing phosphoinositides to total tissue mass are known to be densely populated diacylglycerols and on their subsequent with CB 1 receptors. Peripheral tissues in which transformation to 2-arachidonylglycerol by the CB 1 receptors are expressed on neurons include action of two diacylglycerol lipase (DAGL) isozymes, the heart, vas deferens, urinary bladder and small DAGL a and DAGL ß . 23,24 Following their synthesis and intestine. 10,13 release, these endocannabinoids are removed from their sites of action by cellular uptake and degraded Both CB 1 and CB 2 receptors are coupled through by enzymes,
2-arachidonylglycerol mainly by G i/o proteins, negatively to adenylyl cyclase and monoacylglycerol lipase (MAGL) but also by fatty acid positively to mitogen-activated protein kinase. 1,14 In amide hydrolase (FAAH), and anandamide by FAAH addition, CB 1 receptors are coupled to ion channels and/or by palmitoylethanolamide-preferring acid through G i/o proteins, positively toA-type and inwardly amidase (PAA), cyclooxygenase-2, lipoxygenases rectifying potassium channels and negatively to and cytochrome P450. 5,23-25 Other ligands that may N-type and P/Q-type calcium channels. 1,13,14 CB 1 be endocannabinoids are 2-arachidonylglyceryl receptors can also couple to G s proteins to activate ether (noladin ether), O -arachidonoyl ethanolamine adenylyl cyclase, 15-17 the extent to which this occurs
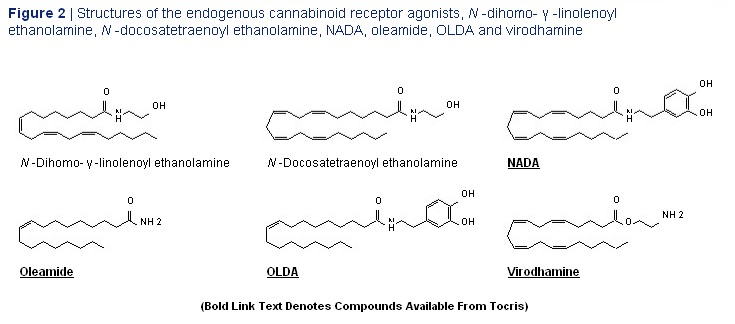
(virodhamine), N -dihomo- ? -linolenoyl ethanolamine, possibly being determined by the location of these N -docosatetraenoyl ethanolamine, oleamide, receptors or by cross-talk between CB 1 receptors N -arachidonoyl dopamine
(NADA) and and co-localised G-protein-coupled non-CB 1 N -oleoyl dopamine (OLDA) (Figures 2 and 3). 5 receptors. 15,16,18,19 It may also be that CB 1 receptors Endocannabinoids together with their receptors can exist as two distinct subpopulations, one constitute what is now usually referred to as the coupled to G i/o proteins and the other to G s . 15 Details `endocannabinoid system'. of additional signalling mechanisms that have been proposed for cannabinoid CB 1 and CB 2 receptors While it is generally accepted that endocannabinoids can be found elsewhere. 1,14 do pass through cell membranes, one issue that is currently very much a matter of debate is the question The cloning of cannabinoid receptors was followed of whether the cellular uptake of endocannabinoids by the discovery that mammalian tissues produce such as anandamide is mediated by a transporter. 25-27 compounds that can activate these receptors. The first In contrast, FAAH is now well characterised. Indeed,
it has been cloned 28 and FAAH knockout mice mechanism(s) underlying the entourage effect have have been developed. 29,30 NAPE/PLD, 31 MAGL, 32-34 yet to be established. and DAGL a and DAGL ß 35 have also been cloned, Endocannabinoids most probably have both and mice with a genetic deletion of NAPE/PLD neuromodulatory and immunomodulatory roles that generated. 36 include inhibition of ongoing transmitter release At least some effects induced by endogenously through retrograde signalling 38 and regulation of released anandamide and 2-arachidonylglycerol cytokine release and of immune cell migration. 39,40 appear to be enhanced through what has been It is also now generally accepted that there are termed the "entourage effect". This relies on certain disorders in which endocannabinoid release the co-release of other endogenous fatty acid increases in particular tissues, and secondly, that this derivatives that include palmitoylethanolamide upregulation of the endocannabinoid system leads and oleamide, which can potentiate anandamide, in some instances to the suppression of unwanted and
2-linoleoylglycerol and
2-palmitoylglycerol, signs and symptoms and so is "autoprotective" and in which can potentiate 2-arachidonylglycerol. 37 The others to the production of undesirable effects. 5 Thus for example, there is evidence that
ND, not determined; THC, tetrahydrocannabinol. See Figure 1 for the structures
of the compounds listed in this table. For further information see references 1, 2 and 41.

endocannabinoid tetrahydrocannabinol ( ? 9 -THC), (-)-11-hydroxy- ? 8 - release on the one hand ameliorates spasticity in THC-dimethylheptyl (HU 210) and CP 55,940 multiple sclerosis and inflammatory pain and on (Table 1 and Figure 1). As to the aminoalkylindole, the other hand contributes towards obesity in some WIN 55,212, whilst its ( R )-(+)-isomer (WIN 55,212-2) individuals or impairs fertility in certain women. As exhibits significant agonist activity at both CB 1 and CB 2 a result, there is now enormous interest not only in receptors, its ( S )-(-)-isomer (WIN 55,212-3 ) does not. directly acting cannabinoid receptor agonists and Indeed, when administered in vitro at concentrations antagonists but also in compounds that can affect the in the low micromolar range, WIN 55,212-3 has been activity of the endocannabinoid system indirectly by found to behave as a partial inverse agonist at CB 1 allosterically modulating endocannabinoid-induced receptors and as a neutral CB 2 receptor antagonist. 44 activation of cannabinoid receptors or by altering the The eicosanoid cannabinoid, anandamide, does concentration of endocannabinoids at their receptors not contain any chiral centres. However, some of through effects on endocannabinoid production or its synthetic analogues do, one example being fate. The remainder of this review describes the the CB 1 -selective agonist, methanandamide (see main pharmacological actions of a number of such next section), the ( R )-(+)-isomer of which has nine direct and indirect cannabinoid receptor agonists times greater affinity for CB 1 receptors than the and antagonists. It focuses particularly on those compounds that are most widely used in cannabinoid CP 55,940, Potent CB 1 and CB 2 research as experimental tools. Whenever possible, Agonist previous review articles have been cited that OH provide more detailed information and list additional CP 55,940 references. Cat. No. 0949 OH OH Mixed CB 1 /CB 2 Receptor Agonists As has been detailed elsewhere, 1,2,41 compounds Me Me that are known to activate CB 1 and CB 2 receptors CP 55,940 is a cannabinoid agonist that is considerably with approximately equal potency and that are more potent than ? 9 -THC in both behavioural tests and most commonly used in the laboratory as CB 1 /CB 2 receptor binding assays. It displays high and roughly equal receptor agonists fall essentially into one of four affinity for both central and peripheral cannabinoid receptors chemical groups: classical cannabinoid, nonclassical
(K i = 0.5-5.0 and 0.69-2.8 nM at CB 1 and CB 2 receptors cannabinoid, aminoalkylindole and eicosanoid respectively).
(Table 1 and Figure 1). Wiley et al (1995) Discriminative stimulus effects of CP 55,940 and structurally dissimilar cannabinoids in rats. Neuropharmacology 34 669. Gatley et al (1997) Many widely used CB 1 /CB 2 receptor agonists contain Binding of the non-classical cannabinoid CP 55,940, and the diarylpyrazole chiral centres and generally exhibit signs of marked AM251 to rodent brain cannabinoid receptors. Life Sci. 61 191. Griffin et al (1998) Evaluation of cannabinoid receptor agonists and antagonists using stereoselectivity in pharmacological assays in which the guanosine-5'-O-(3-[ 35 S]thio)-triphosphate binding assay in rat cerebellar the measured response is CB 1 or CB 2 receptor- membranes. J. Pharmacol. Exp. Ther. 285 553. Thomas et al (1998) Comparative mediated. 1,2,41 Usually, (-)- trans (6a R , 10a R ) classical receptor binding analyses of cannabinoid agonists and antagonists. J. Pharmacol. Exp. Ther. 285 285. and nonclassical cannabinoids exhibit significantly (Canadian customers require a CDSA import permit) greater potency as cannabinoid receptor agonists than their (+)- cis (6a S , 10a S ) enantiomers, three notable examples of such compounds being (-)- ? 9 - for example, there is evidence that endocannabinoid tetrahydrocannabinol ( ? 9 -THC), (-)-11-hydroxy- ? 8 - release on the one hand ameliorates spasticity in THC-dimethylheptyl (HU 210) and CP 55,940 multiple sclerosis and inflammatory pain and on (Table 1 and Figure 1). As to the aminoalkylindole, the other hand contributes towards obesity in some WIN 55,212, whilst its ( R )-(+)-isomer (WIN 55,212-2) individuals or impairs fertility in certain women. As exhibits significant agonist activity at both CB 1 and CB 2 a result, there is now enormous interest not only in receptors, its ( S )-(-)-isomer (WIN 55,212-3 ) does not. directly acting cannabinoid receptor agonists and Indeed, when administered in vitro at concentrations antagonists but also in compounds that can affect the in the low micromolar range, WIN 55,212-3 has been activity of the endocannabinoid system indirectly by found to behave as a partial inverse agonist at CB 1 allosterically modulating endocannabinoid-induced receptors and as a neutral CB 2 receptor antagonist. 44 activation of cannabinoid receptors or by altering the The eicosanoid cannabinoid, anandamide, does concentration of endocannabinoids at their receptors not contain any chiral centres. However, some of through effects on endocannabinoid production or its synthetic analogues do, one example being fate. The remainder of this review describes the the CB 1 -selective agonist, methanandamide (see main pharmacological actions of a number of such next section), the ( R )-(+)-isomer of which has nine direct and indirect cannabinoid receptor agonists times greater affinity for CB 1 receptors than the and antagonists. It focuses particularly on those ( S )-(-)-isomer. 45
One major practical difficulty associated with cannabinoid research, both in vivo and in vitro , is JWH 133, Potent and Selective CB 2 the high lipophilicity and low water solubility of most CB 1 and CB 2 receptor ligands as this necessitates Receptor Agonist Me Me the use of a non-aqueous vehicle such as ethanol, JWH 133 dimethyl sulphoxide, polyvinylpyrrolidone, Tween 80, H Cat. No. 1343 Me Cremophor, Emulphor, bovine serum albumin O or the water-soluble emulsion Tocrisolve 100, H which is a mixture of soya oil, Pluronic F68 and Me Me water. 1,46,47 Consequently, one other cannabinoid JWH 133 is a potent CB 2 agonist that displays approximately CB 1 /CB 2 receptor agonist that merits special 200-fold selectivity over CB 1 receptors (K i values are 3.4 and mention is the Organix compound, 3-(5'-cyano- 677 nM respectively). In vivo , JWH 133 reduces spasticity 1',1'-dimethylpentyl)-1-(4-N-morpholinobutyryloxy)- in a murine autoimmune model of multiple sclerosis. The ? 8 -THC hydrochloride (O-1057), 48 as this is readily superior selectivity, potency and in vivo activity of this CB 2 soluble in water. The in vitro potency of O-1057 agonist make it an important and essential tool for studying relative to that of CP 55,940 is just 2.9 times less at the physiological function of CB 2 receptors. Huffman et al (1999) 3-(1'-Dimethylbutyl)-1-deoxy- ? 8 -THC and related CB 1 receptors and 6.5 times less at CB 2 receptors. compounds: synthesis of selective ligands for the CB 2 receptor. Bioorg. Med. Chem. 7 2905. Pertwee (1999) Pharmacology of cannabinoid receptor ligands. CB 1 -Selective Agonists Curr. Med. Chem. 6 635. Baker et al (2000) Cannabinoids control spasticity and For the development of the first CB 1 -selective tremor in a multiple sclerosis model. Nature 404 84. agonists, the starting point was the anandamide
(DEA controlled substance. Please consult your local molecule, the marginal CB 1 selectivity of which can office for further information. Canadian customers require a CDSA import permit) be significantly enhanced by inserting a fluorine atom on the terminal 2' carbon to form O-585 and/or by mediated hydrolysis. 51

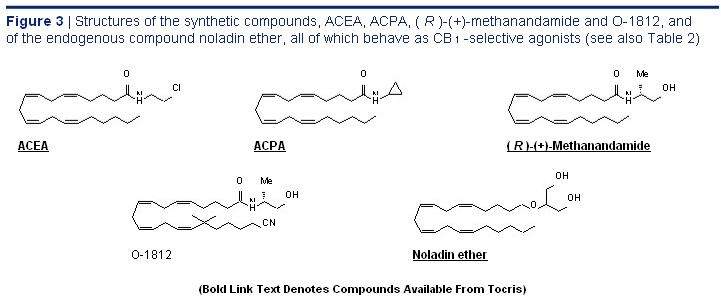
This structural change also replacing a hydrogen atom on the 1' or 2 carbon with reduces the affinity of ACEA for CB 1 receptors by a methyl group to form ( R )-(+)-Methanandamide about 14-fold. One other arachidonic acid derivative its cyano analogue O-1812, or O-689. 1,2,41 Another that deserves mention as a CB 1 -selective agonist is important consequence of inserting a methyl
2-arachidonylglyceryl ether (noladin ether), not least group on the 1' or 2 carbon is greater resistance because it is a putative endocannabinoid. 52 This to the hydrolytic action of FAAH and, indeed, ligand exhibits CP 55,940-like CB 1 efficacy but less
( R )-(+)-Methanandamide was first synthesised CB 1 potency than CP 55,940. 53,54 The structures of in Dr Alexandros Makriyannis' laboratory in order
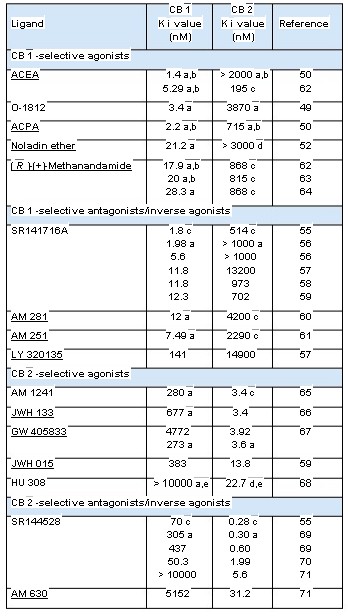
( R )-(+)-Methanandamide, O-1812, ACEA, ACPA and to meet the need for a metabolically more stable noladin ether are shown in Figure 3 and the CB 1 anandamide analogue. Together with O-1812, 49 the and CB 2 binding properties of these compounds are most potent CB 1 -selective agonists so far developed summarised in Table 2. have been arachidonyl-2'-chloroethylamide (ACEA) and arachidonylcyclopropylamide (ACPA), both of CB 2 -Selective Agonists which exhibit reasonably high CB 1 efficacy. 50 However, The CB 2 -selective agonists most widely used unlike O-1812, or indeed methanandamide or O-689, as experimental tools have been the classical neither ACEA nor ACPA show any sign of resistance to cannabinoid, JWH 133, and the less selective enzymic hydrolysis. 1,2,49 This is presumably because aminoalkylindole, JWH 133, both developed by they lack a methyl substituent on the 1' or 2 carbon Dr John Huffman. 1,2,72 Each of these agents not only and, indeed, it has been shown that the addition of a binds more readily to CB 2 than to CB 1 receptors methyl group to the 1' carbon of ACEA does markedly but also behaves as a potent CB 2 -selective agonist decrease the susceptibility of this molecule to FAAH-mediated hydrolysis. 51 This structural change also replacing a hydrogen atom on the 1' or 2 carbon with reduces the affinity of ACEA for CB 1 receptors by a methyl group to form ( R )-(+)-Methanandamide about 14-fold. One other arachidonic acid derivative its cyano analogue O-1812, or O-689. 1,2,41 Another that deserves mention as a CB 1 -selective agonist is important consequence of inserting a methyl
2-arachidonylglyceryl ether (noladin ether), not least group on the 1' or 2 carbon is greater resistance because it is a putative endocannabinoid. 52 This to the hydrolytic action of FAAH and, indeed, ligand exhibits CP 55,940-like CB 1 efficacy but less
( R )-(+)-Methanandamide was first synthesised CB 1 potency than CP 55,940. 53,54 The structures of in Dr Alexandros Makriyannis' laboratory in order
( R )-(+)-Methanandamide, O-1812, ACEA, ACPA and to meet the need for a metabolically more stable noladin ether are shown in Figure 3 and the CB 1 anandamide analogue. Together with O-1812, 49 the and CB 2 binding properties of these compounds are most potent CB 1 -selective agonists so far developed summarised in Table 2.
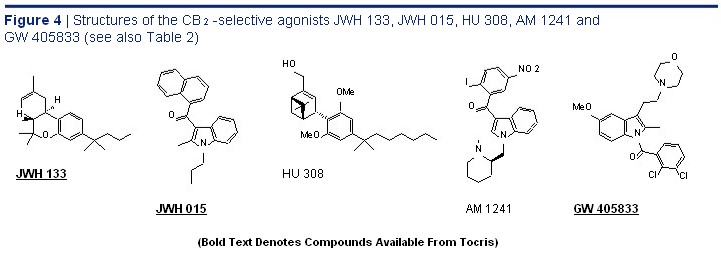
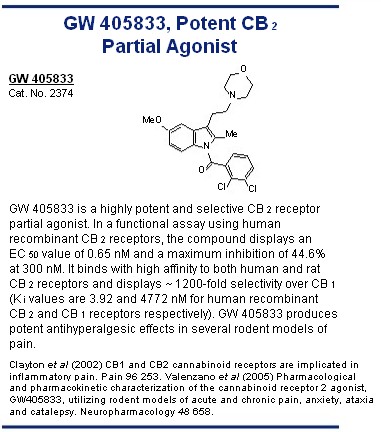
agonists include the GlaxoSmithKline compound GW 405833, which behaves as a potent partial GW 405833, Potent CB 2 agonist at the CB 2 receptor, 67 and HU 308, AM 1241 and the Merck Frosst compounds L-759,633 and Partial Agonist O L-759,656. 1,2 Interestingly, AM 1241 may be a GW 405833 "protean agonist" as it has been reported to behave Cat. No. 2374 N as an agonist in tissues in which CB 2 receptors

are naturally expressed but not in tissues in which CB 2 receptors have been inserted genetically and are therefore presumably overexpressed. 73 The structures of JWH 133, JWH 015, HU 308, AM 1241 and GW 405833 are shown in Figure 4 and their CB 1 and CB 2 binding properties are summarised in Table 2.
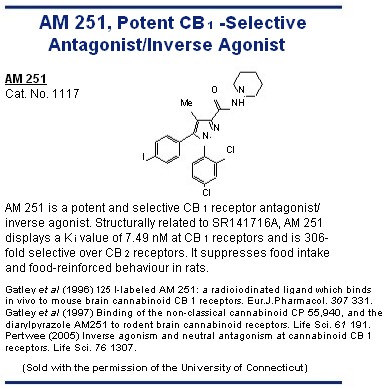
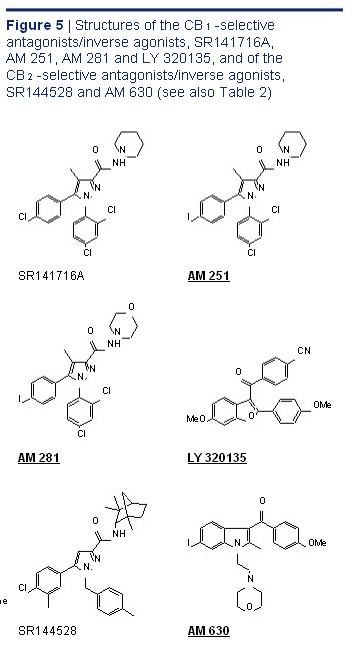
Selective CB 1 Receptor Antagonists/ CB 2 -selective agonists Inverse Agonists The first of these to be developed was the diarylpyrazole, SR141716A. 56 This is a highly potent and selective CB 1 receptor ligand that readily prevents or reverses CB 1 -mediated effects both in vitro and in vivo . 1,2,41 Other notable CB 1 -selective antagonists are AM 251 and AM 281, both developed CB 2 -selective antagonists/inverse agonists by Dr Alexandros Makriyannis, and LY 320135 which has less affinity for CB 1 receptors than SR141716A, AM 251 or AM 281 and at concentrations in the low micromolar range also binds to muscarinic and
5-hydroxytryptamine (5-HT 2 ) receptors. 1,2,41 As detailed elsewhere, 2,74 there is convincing evidence that SR141716A, AM 251, AM 281 and
(Bold Text Denotes Compounds Available From Tocris) LY 320135 are not "neutral" antagonists. Thus, as ACEA, arachidonyl-2'-chloroethylamide; ACPA, arachidonylcyclopropylamide. well as attenuating effects of CB 1 receptor agonists, a Binding to rat cannabinoid receptors in transfected cells or in brain (mainly CB 1 ) they can by themselves elicit responses in some or spleen tissue (mainly CB 2 ). b With phenylmethylsulphonyl fluoride in order to inhibit enzymic hydrolysis. CB 1 receptor-containing tissues that are opposite in c Binding to mouse brain (mainly CB 1 ) or spleen tissue (mainly CB 2 ). direction from those elicited by CB 1 receptor agonists. d Species unspecified. Whilst such "inverse cannabimimetic effects" All other data from experiments with human cannabinoid receptors. may in some instances be attributable to a direct antagonism of responses evoked at CB 1 receptors e Displacement of [ 3 H]HU 243 from CB 1 - and CB 2 -specific binding sites; [ 3 H]CP 55,940 was used in all other experiments. by released endocannabinoids, there is evidence that this is not always the underlying mechanism and See Figures 3 to 5 for the structures of the compounds listed in this table. that SR141716A, AM 251, AM 281 and LY 320135 are in fact inverse agonists. 74 More specifically, they appear to produce inverse cannabimimetic effects LY 320135, CB 1 Antagonist/ in at least some tissues by somehow reducing the constitutive activity of CB 1 receptors (the coupling of Inverse Agonist CN CB 1 receptors to their effector mechanisms that, it is LY 320135 O thought, can occur in the absence of exogenously Cat. No. 2387 added or endogenously released CB 1 agonists). OMe The structures of SR141716A, AM 251, AM 281 MeO O and LY 320135 are shown in Figure 5 and the CB 1 and CB 2 binding properties of these compounds are LY 320135 is a CB 1 receptor antagonist that is structurally summarised in Table 2. dissimilar from SR 141716A and AM 251. The compound produces inverse agonist effects and displays 70-fold Selective CB 2 Receptor Antagonists/ selectivity for CB 1 over CB 2 receptors (K i values are 141 nM Inverse Agonists and 10 µ M respectively. It shows weak binding to both
5-HT 2 (K i = 6.4 µ M) and muscarinic receptors (K i = 2.1 µ M). The most notable CB 2 -selective antagonists/inverse Felder et al (1998) LY320135, a novel cannabinoid CB 1 receptor antagonist, agonists are the Sanofi-Aventis diarylpyrazole, unmasks coupling of the CB 1 receptor to stimulation of cAMP accumulation. SR144528, 69 and 6-iodopravadoline (AM 630) 71 J. Pharmacol. Exp. Ther. 284 291. Holland et al (1999) Cannabinoid CB 1 receptors fail to cause relaxation, but couple via G i /G o to the inhibition of
(Figure 5). Both compounds bind with much higher adenylyl cyclase in carotid artery smooth muscle. Br. J. Pharmacol. 128 597. affinity to CB 2 than to CB 1 receptors (Table 2), exhibit Pertwee (2005) Inverse agonism and neutral antagonism at cannabinoid CB 1 marked potency as CB 2 receptor antagonists and receptors. Life Sci. 76 1307. behave as inverse agonists that can by themselves produce inverse cannabimimetic effects at CB 2 Figure 5 Structures of the CB 1 -selective receptors. 1,2,41 Thus for example, AM 630 has been antagonists/inverse agonists, SR141716A, reported to reverse CP 55,940-induced inhibition AM 251, AM 281 and LY 320135, and of the of forskolin-stimulated cyclic AMP production by CB 2 -selective antagonists/inverse agonists, human CB 2 -transfected CHO cell preparations SR144528 and AM 630 (see also Table 2) at concentrations in the nanomolar range
(EC 50 = 129 nM) and to enhance forskolin-stimulated cyclic AMP production by the same cell line when O N O N administered by itself (EC 50 = 230 nM), 71 albeit with NH NH an efficacy that appears to be somewhat less than N N the inverse efficacy displayed by SR144528 in this N N Cl Cl bioassay. 75 At the CB 1 receptor, AM 630 has been Cl I found to behave in some investigations as a low- potency partial agonist 41,71,76-78 but in others as a low- Cl Cl potency inverse agonist. 79,80

Neutral Cannabinoid Receptor ? 8 -THC with an acetylenic side chain, and VCHR, an Antagonists analogue of SR141716A, are neutral CB 1 receptor There is currently considerable interest in the antagonists. 2,74 As this evidence is somewhat possibility of developing potent neutral CB 1 and CB 2 preliminary, particular caution should be exercised receptor antagonists: i. e. high-affinity ligands for when using any of these ligands as a pharmacological CB 1 or CB 2 receptors that lack significant agonist or tool. There is more complete evidence that another inverse agonist efficacy. One reason for this is that SR141716A analogue, NESS 0327, is a neutral CB 1 unlike the CB 1 and CB 2 -selective antagonists/inverse receptor antagonist. 55 However, this compound is agonists now available (see previous sections), currently not commercially available and so has not a neutral antagonist could be used to distinguish been much used in cannabinoid research. between tonic cannabimimetic activity arising from Radiolabelled Cannabinoid Receptor ongoing endocannabinoid release onto CB 1 or CB 2 receptors, which it should oppose, and tonic activity Ligands arising from the presence of constitutively active CB 1 Tritiated cannabinoid receptor ligands that or CB 2 receptors, which it should not. Although no have been most widely used in binding assays neutral antagonist that selectively targets the CB 2 or for autoradiography are the CB 1 -selective receptor has yet been developed, some progress has [ 3 H]SR141716A (CB 1 K d = 0.19 to 1.24 nM), and been made on the CB 1 receptor front. Thus, there is [ 3 H]CP 55,940, [ 3 H]WIN 55,212-2 and [ 3 H]HU 243, some evidence that 6''-azidohex-2''-yne-cannabidiol all three of which bind more or less equally well (O-2654), O-2050, a sulphonamide analogue of ? 8 -THC with an acetylenic side chain, and VCHR, an Antagonists analogue of SR141716A, are neutral CB 1 receptor There is currently considerable interest in the antagonists. 2,74 As this evidence is somewhat possibility of developing potent neutral CB 1 and CB 2 preliminary, particular caution should be exercised receptor antagonists: i. e. high-affinity ligands for when using any of these ligands as a pharmacological CB 1 or CB 2 receptors that lack significant agonist or tool. There is more complete evidence that another inverse agonist efficacy. One reason for this is that SR141716A analogue, NESS 0327, is a neutral CB 1 unlike the CB 1 and CB 2 -selective antagonists/inverse receptor antagonist. 55 However, this compound is agonists now available (see previous sections), currently not commercially available and so has not a neutral antagonist could be used to distinguish been much used in cannabinoid research. between tonic cannabimimetic activity arising from Radiolabelled Cannabinoid Receptor ongoing endocannabinoid release onto CB 1 or CB 2 receptors, which it should oppose, and tonic activity Ligands arising from the presence of constitutively active CB 1 Tritiated cannabinoid receptor ligands that or CB 2 receptors, which it should not. Although no have been most widely used in binding assays neutral antagonist that selectively targets the CB 2 or for autoradiography are the CB 1 -selective receptor has yet been developed, some progress has [ 3 H]SR141716A (CB 1 K d = 0.19 to 1.24 nM), and been made on the CB 1 receptor front. Thus, there is [ 3 H]CP 55,940, [ 3 H]WIN 55,212-2 and [ 3 H]HU 243, some evidence that 6''-azidohex-2''-yne-cannabidiol all three of which bind more or less equally well (O-2654), O-2050, a sulphonamide analogue of to CB 1 and CB 2 receptors. Typical K d values for
Roger G Pertwee School of Medical Sciences, Institute of Medical Sciences, University of Aberdeen, Foresterhill, Aberdeen
Roger Pertwee is currently Professor of Neuropharmacology at the University of Aberdeen and Director of Pharmacology for GW Pharmaceuticals. His research focuses on thepharmacology of cannabis and its constituents and of cannabinoid receptors and cannabis-derived, synthetic and endogenous ligands for these receptors.
The Endocannabinoid System including immune cells. CB 2 receptors are located Two types of cannabinoid receptor have so far been predominantly in immune cells both within and identified. 1,2 These are the CB 1 receptor, cloned outside the central nervous system, the functions in 1990, 3 and the CB 2 receptor, cloned in 1993, 4 of these receptors including modulation of cytokine both of which are members of the superfamily of release and of immune cell migration. In the brain, G-protein-coupled receptors. The cloning of these CB 2 receptors are expressed by microglia, 7 by blood receptors prompted the development of mice from vessels, 7 and by some neurons. 8,9 However, the role which cannabinoid CB 1 and/or CB 2 receptors have of neuronal CB 2 receptors is currently unknown. been genetically deleted and these transgenic The central distribution pattern of CB 1 receptors is animals, particularly CB 1 knockout mice, are now heterogeneous and accounts for several prominent widely used to explore the physiological and pharmacological properties of CB 1 receptor agonists, pathological functions of cannabinoid receptors. 1,5,6 for example their ability to impair cognition and CB 1 receptors are found mainly at the terminals of memory and to alter the control of motor function. Thus central and peripheral neurons where they usually the cerebral cortex, hippocampus, lateral caudate- mediate inhibition of neurotransmitter release. putamen, substantia nigra pars reticulata, globus They are also present in some non-neuronal cells, pallidus, entopeduncular nucleus and the molecular

layer of the cerebellum are all populated with such endogenous cannabinoids (endocannabinoids) particularly high concentrations of CB 1 receptors. 1,10 to be identified were N -arachidonoyl ethanolamine In line with the analgesic properties of cannabinoid (anandamide) in 1992 and 2-arachidonylglycerol in receptor agonists, CB 1 receptors are also found on 1995 (Figure 1), 20-22 both of which are synthesised pain pathways in the brain and spinal cord and at the on demand in response to elevations of intracellular peripheral terminals of primary sensory neurons. 11,12 calcium. 23 Anandamideisformedfrom N -arachidonoyl Although the concentration of CB 1 receptors is phosphatidylethanolamine in a process that is considerably less in peripheral tissues than in the catalysed by N -acyl phosphatidylethanolamine- central nervous system, this does not mean that selective phospholipase D (NAPE-PLD). The peripheral CB 1 receptors are unimportant. Thus in synthesis of
2-arachidonylglycerol, however, some peripheral tissues, discrete regions such as is thought to depend on the conversion of nerve terminals that form only a small part of the
2-arachidonate-containing phosphoinositides to total tissue mass are known to be densely populated diacylglycerols and on their subsequent with CB 1 receptors. Peripheral tissues in which transformation to 2-arachidonylglycerol by the CB 1 receptors are expressed on neurons include action of two diacylglycerol lipase (DAGL) isozymes, the heart, vas deferens, urinary bladder and small DAGL a and DAGL ß . 23,24 Following their synthesis and intestine. 10,13 release, these endocannabinoids are removed from their sites of action by cellular uptake and degraded Both CB 1 and CB 2 receptors are coupled through by enzymes,
2-arachidonylglycerol mainly by G i/o proteins, negatively to adenylyl cyclase and monoacylglycerol lipase (MAGL) but also by fatty acid positively to mitogen-activated protein kinase. 1,14 In amide hydrolase (FAAH), and anandamide by FAAH addition, CB 1 receptors are coupled to ion channels and/or by palmitoylethanolamide-preferring acid through G i/o proteins, positively toA-type and inwardly amidase (PAA), cyclooxygenase-2, lipoxygenases rectifying potassium channels and negatively to and cytochrome P450. 5,23-25 Other ligands that may N-type and P/Q-type calcium channels. 1,13,14 CB 1 be endocannabinoids are 2-arachidonylglyceryl receptors can also couple to G s proteins to activate ether (noladin ether), O -arachidonoyl ethanolamine adenylyl cyclase, 15-17 the extent to which this occurs
(virodhamine), N -dihomo- ? -linolenoyl ethanolamine, possibly being determined by the location of these N -docosatetraenoyl ethanolamine, oleamide, receptors or by cross-talk between CB 1 receptors N -arachidonoyl dopamine
(NADA) and and co-localised G-protein-coupled non-CB 1 N -oleoyl dopamine (OLDA) (Figures 2 and 3). 5 receptors. 15,16,18,19 It may also be that CB 1 receptors Endocannabinoids together with their receptors can exist as two distinct subpopulations, one constitute what is now usually referred to as the coupled to G i/o proteins and the other to G s . 15 Details `endocannabinoid system'. of additional signalling mechanisms that have been proposed for cannabinoid CB 1 and CB 2 receptors While it is generally accepted that endocannabinoids can be found elsewhere. 1,14 do pass through cell membranes, one issue that is currently very much a matter of debate is the question The cloning of cannabinoid receptors was followed of whether the cellular uptake of endocannabinoids by the discovery that mammalian tissues produce such as anandamide is mediated by a transporter. 25-27 compounds that can activate these receptors. The first In contrast, FAAH is now well characterised. Indeed,

it has been cloned 28 and FAAH knockout mice mechanism(s) underlying the entourage effect have have been developed. 29,30 NAPE/PLD, 31 MAGL, 32-34 yet to be established. and DAGL a and DAGL ß 35 have also been cloned, Endocannabinoids most probably have both and mice with a genetic deletion of NAPE/PLD neuromodulatory and immunomodulatory roles that generated. 36 include inhibition of ongoing transmitter release At least some effects induced by endogenously through retrograde signalling 38 and regulation of released anandamide and 2-arachidonylglycerol cytokine release and of immune cell migration. 39,40 appear to be enhanced through what has been It is also now generally accepted that there are termed the "entourage effect". This relies on certain disorders in which endocannabinoid release the co-release of other endogenous fatty acid increases in particular tissues, and secondly, that this derivatives that include palmitoylethanolamide upregulation of the endocannabinoid system leads and oleamide, which can potentiate anandamide, in some instances to the suppression of unwanted and
2-linoleoylglycerol and
2-palmitoylglycerol, signs and symptoms and so is "autoprotective" and in which can potentiate 2-arachidonylglycerol. 37 The others to the production of undesirable effects. 5 Thus for example, there is evidence that

ND, not determined; THC, tetrahydrocannabinol. See Figure 1 for the structures
of the compounds listed in this table. For further information see references 1, 2 and 41.

endocannabinoid tetrahydrocannabinol ( ? 9 -THC), (-)-11-hydroxy- ? 8 - release on the one hand ameliorates spasticity in THC-dimethylheptyl (HU 210) and CP 55,940 multiple sclerosis and inflammatory pain and on (Table 1 and Figure 1). As to the aminoalkylindole, the other hand contributes towards obesity in some WIN 55,212, whilst its ( R )-(+)-isomer (WIN 55,212-2) individuals or impairs fertility in certain women. As exhibits significant agonist activity at both CB 1 and CB 2 a result, there is now enormous interest not only in receptors, its ( S )-(-)-isomer (WIN 55,212-3 ) does not. directly acting cannabinoid receptor agonists and Indeed, when administered in vitro at concentrations antagonists but also in compounds that can affect the in the low micromolar range, WIN 55,212-3 has been activity of the endocannabinoid system indirectly by found to behave as a partial inverse agonist at CB 1 allosterically modulating endocannabinoid-induced receptors and as a neutral CB 2 receptor antagonist. 44 activation of cannabinoid receptors or by altering the The eicosanoid cannabinoid, anandamide, does concentration of endocannabinoids at their receptors not contain any chiral centres. However, some of through effects on endocannabinoid production or its synthetic analogues do, one example being fate. The remainder of this review describes the the CB 1 -selective agonist, methanandamide (see main pharmacological actions of a number of such next section), the ( R )-(+)-isomer of which has nine direct and indirect cannabinoid receptor agonists times greater affinity for CB 1 receptors than the and antagonists. It focuses particularly on those compounds that are most widely used in cannabinoid CP 55,940, Potent CB 1 and CB 2 research as experimental tools. Whenever possible, Agonist previous review articles have been cited that OH provide more detailed information and list additional CP 55,940 references. Cat. No. 0949 OH OH Mixed CB 1 /CB 2 Receptor Agonists As has been detailed elsewhere, 1,2,41 compounds Me Me that are known to activate CB 1 and CB 2 receptors CP 55,940 is a cannabinoid agonist that is considerably with approximately equal potency and that are more potent than ? 9 -THC in both behavioural tests and most commonly used in the laboratory as CB 1 /CB 2 receptor binding assays. It displays high and roughly equal receptor agonists fall essentially into one of four affinity for both central and peripheral cannabinoid receptors chemical groups: classical cannabinoid, nonclassical
(K i = 0.5-5.0 and 0.69-2.8 nM at CB 1 and CB 2 receptors cannabinoid, aminoalkylindole and eicosanoid respectively).
(Table 1 and Figure 1). Wiley et al (1995) Discriminative stimulus effects of CP 55,940 and structurally dissimilar cannabinoids in rats. Neuropharmacology 34 669. Gatley et al (1997) Many widely used CB 1 /CB 2 receptor agonists contain Binding of the non-classical cannabinoid CP 55,940, and the diarylpyrazole chiral centres and generally exhibit signs of marked AM251 to rodent brain cannabinoid receptors. Life Sci. 61 191. Griffin et al (1998) Evaluation of cannabinoid receptor agonists and antagonists using stereoselectivity in pharmacological assays in which the guanosine-5'-O-(3-[ 35 S]thio)-triphosphate binding assay in rat cerebellar the measured response is CB 1 or CB 2 receptor- membranes. J. Pharmacol. Exp. Ther. 285 553. Thomas et al (1998) Comparative mediated. 1,2,41 Usually, (-)- trans (6a R , 10a R ) classical receptor binding analyses of cannabinoid agonists and antagonists. J. Pharmacol. Exp. Ther. 285 285. and nonclassical cannabinoids exhibit significantly (Canadian customers require a CDSA import permit) greater potency as cannabinoid receptor agonists than their (+)- cis (6a S , 10a S ) enantiomers, three notable examples of such compounds being (-)- ? 9 - for example, there is evidence that endocannabinoid tetrahydrocannabinol ( ? 9 -THC), (-)-11-hydroxy- ? 8 - release on the one hand ameliorates spasticity in THC-dimethylheptyl (HU 210) and CP 55,940 multiple sclerosis and inflammatory pain and on (Table 1 and Figure 1). As to the aminoalkylindole, the other hand contributes towards obesity in some WIN 55,212, whilst its ( R )-(+)-isomer (WIN 55,212-2) individuals or impairs fertility in certain women. As exhibits significant agonist activity at both CB 1 and CB 2 a result, there is now enormous interest not only in receptors, its ( S )-(-)-isomer (WIN 55,212-3 ) does not. directly acting cannabinoid receptor agonists and Indeed, when administered in vitro at concentrations antagonists but also in compounds that can affect the in the low micromolar range, WIN 55,212-3 has been activity of the endocannabinoid system indirectly by found to behave as a partial inverse agonist at CB 1 allosterically modulating endocannabinoid-induced receptors and as a neutral CB 2 receptor antagonist. 44 activation of cannabinoid receptors or by altering the The eicosanoid cannabinoid, anandamide, does concentration of endocannabinoids at their receptors not contain any chiral centres. However, some of through effects on endocannabinoid production or its synthetic analogues do, one example being fate. The remainder of this review describes the the CB 1 -selective agonist, methanandamide (see main pharmacological actions of a number of such next section), the ( R )-(+)-isomer of which has nine direct and indirect cannabinoid receptor agonists times greater affinity for CB 1 receptors than the and antagonists. It focuses particularly on those ( S )-(-)-isomer. 45

One major practical difficulty associated with cannabinoid research, both in vivo and in vitro , is JWH 133, Potent and Selective CB 2 the high lipophilicity and low water solubility of most CB 1 and CB 2 receptor ligands as this necessitates Receptor Agonist Me Me the use of a non-aqueous vehicle such as ethanol, JWH 133 dimethyl sulphoxide, polyvinylpyrrolidone, Tween 80, H Cat. No. 1343 Me Cremophor, Emulphor, bovine serum albumin O or the water-soluble emulsion Tocrisolve 100, H which is a mixture of soya oil, Pluronic F68 and Me Me water. 1,46,47 Consequently, one other cannabinoid JWH 133 is a potent CB 2 agonist that displays approximately CB 1 /CB 2 receptor agonist that merits special 200-fold selectivity over CB 1 receptors (K i values are 3.4 and mention is the Organix compound, 3-(5'-cyano- 677 nM respectively). In vivo , JWH 133 reduces spasticity 1',1'-dimethylpentyl)-1-(4-N-morpholinobutyryloxy)- in a murine autoimmune model of multiple sclerosis. The ? 8 -THC hydrochloride (O-1057), 48 as this is readily superior selectivity, potency and in vivo activity of this CB 2 soluble in water. The in vitro potency of O-1057 agonist make it an important and essential tool for studying relative to that of CP 55,940 is just 2.9 times less at the physiological function of CB 2 receptors. Huffman et al (1999) 3-(1'-Dimethylbutyl)-1-deoxy- ? 8 -THC and related CB 1 receptors and 6.5 times less at CB 2 receptors. compounds: synthesis of selective ligands for the CB 2 receptor. Bioorg. Med. Chem. 7 2905. Pertwee (1999) Pharmacology of cannabinoid receptor ligands. CB 1 -Selective Agonists Curr. Med. Chem. 6 635. Baker et al (2000) Cannabinoids control spasticity and For the development of the first CB 1 -selective tremor in a multiple sclerosis model. Nature 404 84. agonists, the starting point was the anandamide
(DEA controlled substance. Please consult your local molecule, the marginal CB 1 selectivity of which can office for further information. Canadian customers require a CDSA import permit) be significantly enhanced by inserting a fluorine atom on the terminal 2' carbon to form O-585 and/or by mediated hydrolysis. 51

This structural change also replacing a hydrogen atom on the 1' or 2 carbon with reduces the affinity of ACEA for CB 1 receptors by a methyl group to form ( R )-(+)-Methanandamide about 14-fold. One other arachidonic acid derivative its cyano analogue O-1812, or O-689. 1,2,41 Another that deserves mention as a CB 1 -selective agonist is important consequence of inserting a methyl
2-arachidonylglyceryl ether (noladin ether), not least group on the 1' or 2 carbon is greater resistance because it is a putative endocannabinoid. 52 This to the hydrolytic action of FAAH and, indeed, ligand exhibits CP 55,940-like CB 1 efficacy but less
( R )-(+)-Methanandamide was first synthesised CB 1 potency than CP 55,940. 53,54 The structures of in Dr Alexandros Makriyannis' laboratory in order
( R )-(+)-Methanandamide, O-1812, ACEA, ACPA and to meet the need for a metabolically more stable noladin ether are shown in Figure 3 and the CB 1 anandamide analogue. Together with O-1812, 49 the and CB 2 binding properties of these compounds are most potent CB 1 -selective agonists so far developed summarised in Table 2. have been arachidonyl-2'-chloroethylamide (ACEA) and arachidonylcyclopropylamide (ACPA), both of CB 2 -Selective Agonists which exhibit reasonably high CB 1 efficacy. 50 However, The CB 2 -selective agonists most widely used unlike O-1812, or indeed methanandamide or O-689, as experimental tools have been the classical neither ACEA nor ACPA show any sign of resistance to cannabinoid, JWH 133, and the less selective enzymic hydrolysis. 1,2,49 This is presumably because aminoalkylindole, JWH 133, both developed by they lack a methyl substituent on the 1' or 2 carbon Dr John Huffman. 1,2,72 Each of these agents not only and, indeed, it has been shown that the addition of a binds more readily to CB 2 than to CB 1 receptors methyl group to the 1' carbon of ACEA does markedly but also behaves as a potent CB 2 -selective agonist decrease the susceptibility of this molecule to FAAH-mediated hydrolysis. 51 This structural change also replacing a hydrogen atom on the 1' or 2 carbon with reduces the affinity of ACEA for CB 1 receptors by a methyl group to form ( R )-(+)-Methanandamide about 14-fold. One other arachidonic acid derivative its cyano analogue O-1812, or O-689. 1,2,41 Another that deserves mention as a CB 1 -selective agonist is important consequence of inserting a methyl
2-arachidonylglyceryl ether (noladin ether), not least group on the 1' or 2 carbon is greater resistance because it is a putative endocannabinoid. 52 This to the hydrolytic action of FAAH and, indeed, ligand exhibits CP 55,940-like CB 1 efficacy but less
( R )-(+)-Methanandamide was first synthesised CB 1 potency than CP 55,940. 53,54 The structures of in Dr Alexandros Makriyannis' laboratory in order
( R )-(+)-Methanandamide, O-1812, ACEA, ACPA and to meet the need for a metabolically more stable noladin ether are shown in Figure 3 and the CB 1 anandamide analogue. Together with O-1812, 49 the and CB 2 binding properties of these compounds are most potent CB 1 -selective agonists so far developed summarised in Table 2.

agonists include the GlaxoSmithKline compound GW 405833, which behaves as a potent partial GW 405833, Potent CB 2 agonist at the CB 2 receptor, 67 and HU 308, AM 1241 and the Merck Frosst compounds L-759,633 and Partial Agonist O L-759,656. 1,2 Interestingly, AM 1241 may be a GW 405833 "protean agonist" as it has been reported to behave Cat. No. 2374 N as an agonist in tissues in which CB 2 receptors



are naturally expressed but not in tissues in which CB 2 receptors have been inserted genetically and are therefore presumably overexpressed. 73 The structures of JWH 133, JWH 015, HU 308, AM 1241 and GW 405833 are shown in Figure 4 and their CB 1 and CB 2 binding properties are summarised in Table 2.
Selective CB 1 Receptor Antagonists/ CB 2 -selective agonists Inverse Agonists The first of these to be developed was the diarylpyrazole, SR141716A. 56 This is a highly potent and selective CB 1 receptor ligand that readily prevents or reverses CB 1 -mediated effects both in vitro and in vivo . 1,2,41 Other notable CB 1 -selective antagonists are AM 251 and AM 281, both developed CB 2 -selective antagonists/inverse agonists by Dr Alexandros Makriyannis, and LY 320135 which has less affinity for CB 1 receptors than SR141716A, AM 251 or AM 281 and at concentrations in the low micromolar range also binds to muscarinic and
5-hydroxytryptamine (5-HT 2 ) receptors. 1,2,41 As detailed elsewhere, 2,74 there is convincing evidence that SR141716A, AM 251, AM 281 and
(Bold Text Denotes Compounds Available From Tocris) LY 320135 are not "neutral" antagonists. Thus, as ACEA, arachidonyl-2'-chloroethylamide; ACPA, arachidonylcyclopropylamide. well as attenuating effects of CB 1 receptor agonists, a Binding to rat cannabinoid receptors in transfected cells or in brain (mainly CB 1 ) they can by themselves elicit responses in some or spleen tissue (mainly CB 2 ). b With phenylmethylsulphonyl fluoride in order to inhibit enzymic hydrolysis. CB 1 receptor-containing tissues that are opposite in c Binding to mouse brain (mainly CB 1 ) or spleen tissue (mainly CB 2 ). direction from those elicited by CB 1 receptor agonists. d Species unspecified. Whilst such "inverse cannabimimetic effects" All other data from experiments with human cannabinoid receptors. may in some instances be attributable to a direct antagonism of responses evoked at CB 1 receptors e Displacement of [ 3 H]HU 243 from CB 1 - and CB 2 -specific binding sites; [ 3 H]CP 55,940 was used in all other experiments. by released endocannabinoids, there is evidence that this is not always the underlying mechanism and See Figures 3 to 5 for the structures of the compounds listed in this table. that SR141716A, AM 251, AM 281 and LY 320135 are in fact inverse agonists. 74 More specifically, they appear to produce inverse cannabimimetic effects LY 320135, CB 1 Antagonist/ in at least some tissues by somehow reducing the constitutive activity of CB 1 receptors (the coupling of Inverse Agonist CN CB 1 receptors to their effector mechanisms that, it is LY 320135 O thought, can occur in the absence of exogenously Cat. No. 2387 added or endogenously released CB 1 agonists). OMe The structures of SR141716A, AM 251, AM 281 MeO O and LY 320135 are shown in Figure 5 and the CB 1 and CB 2 binding properties of these compounds are LY 320135 is a CB 1 receptor antagonist that is structurally summarised in Table 2. dissimilar from SR 141716A and AM 251. The compound produces inverse agonist effects and displays 70-fold Selective CB 2 Receptor Antagonists/ selectivity for CB 1 over CB 2 receptors (K i values are 141 nM Inverse Agonists and 10 µ M respectively. It shows weak binding to both
5-HT 2 (K i = 6.4 µ M) and muscarinic receptors (K i = 2.1 µ M). The most notable CB 2 -selective antagonists/inverse Felder et al (1998) LY320135, a novel cannabinoid CB 1 receptor antagonist, agonists are the Sanofi-Aventis diarylpyrazole, unmasks coupling of the CB 1 receptor to stimulation of cAMP accumulation. SR144528, 69 and 6-iodopravadoline (AM 630) 71 J. Pharmacol. Exp. Ther. 284 291. Holland et al (1999) Cannabinoid CB 1 receptors fail to cause relaxation, but couple via G i /G o to the inhibition of
(Figure 5). Both compounds bind with much higher adenylyl cyclase in carotid artery smooth muscle. Br. J. Pharmacol. 128 597. affinity to CB 2 than to CB 1 receptors (Table 2), exhibit Pertwee (2005) Inverse agonism and neutral antagonism at cannabinoid CB 1 marked potency as CB 2 receptor antagonists and receptors. Life Sci. 76 1307. behave as inverse agonists that can by themselves produce inverse cannabimimetic effects at CB 2 Figure 5 Structures of the CB 1 -selective receptors. 1,2,41 Thus for example, AM 630 has been antagonists/inverse agonists, SR141716A, reported to reverse CP 55,940-induced inhibition AM 251, AM 281 and LY 320135, and of the of forskolin-stimulated cyclic AMP production by CB 2 -selective antagonists/inverse agonists, human CB 2 -transfected CHO cell preparations SR144528 and AM 630 (see also Table 2) at concentrations in the nanomolar range
(EC 50 = 129 nM) and to enhance forskolin-stimulated cyclic AMP production by the same cell line when O N O N administered by itself (EC 50 = 230 nM), 71 albeit with NH NH an efficacy that appears to be somewhat less than N N the inverse efficacy displayed by SR144528 in this N N Cl Cl bioassay. 75 At the CB 1 receptor, AM 630 has been Cl I found to behave in some investigations as a low- potency partial agonist 41,71,76-78 but in others as a low- Cl Cl potency inverse agonist. 79,80



Neutral Cannabinoid Receptor ? 8 -THC with an acetylenic side chain, and VCHR, an Antagonists analogue of SR141716A, are neutral CB 1 receptor There is currently considerable interest in the antagonists. 2,74 As this evidence is somewhat possibility of developing potent neutral CB 1 and CB 2 preliminary, particular caution should be exercised receptor antagonists: i. e. high-affinity ligands for when using any of these ligands as a pharmacological CB 1 or CB 2 receptors that lack significant agonist or tool. There is more complete evidence that another inverse agonist efficacy. One reason for this is that SR141716A analogue, NESS 0327, is a neutral CB 1 unlike the CB 1 and CB 2 -selective antagonists/inverse receptor antagonist. 55 However, this compound is agonists now available (see previous sections), currently not commercially available and so has not a neutral antagonist could be used to distinguish been much used in cannabinoid research. between tonic cannabimimetic activity arising from Radiolabelled Cannabinoid Receptor ongoing endocannabinoid release onto CB 1 or CB 2 receptors, which it should oppose, and tonic activity Ligands arising from the presence of constitutively active CB 1 Tritiated cannabinoid receptor ligands that or CB 2 receptors, which it should not. Although no have been most widely used in binding assays neutral antagonist that selectively targets the CB 2 or for autoradiography are the CB 1 -selective receptor has yet been developed, some progress has [ 3 H]SR141716A (CB 1 K d = 0.19 to 1.24 nM), and been made on the CB 1 receptor front. Thus, there is [ 3 H]CP 55,940, [ 3 H]WIN 55,212-2 and [ 3 H]HU 243, some evidence that 6''-azidohex-2''-yne-cannabidiol all three of which bind more or less equally well (O-2654), O-2050, a sulphonamide analogue of ? 8 -THC with an acetylenic side chain, and VCHR, an Antagonists analogue of SR141716A, are neutral CB 1 receptor There is currently considerable interest in the antagonists. 2,74 As this evidence is somewhat possibility of developing potent neutral CB 1 and CB 2 preliminary, particular caution should be exercised receptor antagonists: i. e. high-affinity ligands for when using any of these ligands as a pharmacological CB 1 or CB 2 receptors that lack significant agonist or tool. There is more complete evidence that another inverse agonist efficacy. One reason for this is that SR141716A analogue, NESS 0327, is a neutral CB 1 unlike the CB 1 and CB 2 -selective antagonists/inverse receptor antagonist. 55 However, this compound is agonists now available (see previous sections), currently not commercially available and so has not a neutral antagonist could be used to distinguish been much used in cannabinoid research. between tonic cannabimimetic activity arising from Radiolabelled Cannabinoid Receptor ongoing endocannabinoid release onto CB 1 or CB 2 receptors, which it should oppose, and tonic activity Ligands arising from the presence of constitutively active CB 1 Tritiated cannabinoid receptor ligands that or CB 2 receptors, which it should not. Although no have been most widely used in binding assays neutral antagonist that selectively targets the CB 2 or for autoradiography are the CB 1 -selective receptor has yet been developed, some progress has [ 3 H]SR141716A (CB 1 K d = 0.19 to 1.24 nM), and been made on the CB 1 receptor front. Thus, there is [ 3 H]CP 55,940, [ 3 H]WIN 55,212-2 and [ 3 H]HU 243, some evidence that 6''-azidohex-2''-yne-cannabidiol all three of which bind more or less equally well (O-2654), O-2050, a sulphonamide analogue of to CB 1 and CB 2 receptors. Typical K d values for



